Loss of T-tubules and other changes to surface topography in ventricular myocytes from failing human and rat heart
- PMID: 19342485
- PMCID: PMC2672472
- DOI: 10.1073/pnas.0809777106
Loss of T-tubules and other changes to surface topography in ventricular myocytes from failing human and rat heart
Abstract
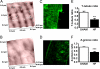
T-tubular invaginations of the sarcolemma of ventricular cardiomyocytes contain junctional structures functionally coupling L-type calcium channels to the sarcoplasmic reticulum calcium-release channels (the ryanodine receptors), and therefore their configuration controls the gain of calcium-induced calcium release (CICR). Studies primarily in rodent myocardium have shown the importance of T-tubular structures for calcium transient kinetics and have linked T-tubule disruption to delayed CICR. However, there is disagreement as to the nature of T-tubule changes in human heart failure. We studied isolated ventricular myocytes from patients with ischemic heart disease, idiopathic dilated cardiomyopathy, and hypertrophic obstructive cardiomyopathy and determined T-tubule structure with either the fluorescent membrane dye di-8-ANNEPs or the scanning ion conductance microscope (SICM). The SICM uses a scanning pipette to produce a topographic representation of the surface of the live cell by a non-optical method. We have also compared ventricular myocytes from a rat model of chronic heart failure after myocardial infarction. T-tubule loss, shown by both ANNEPs staining and SICM imaging, was pronounced in human myocytes from all etiologies of disease. SICM imaging showed additional changes in surface structure, with flattening and loss of Z-groove definition common to all etiologies. Rat myocytes from the chronic heart failure model also showed both T-tubule and Z-groove loss, as well as increased spark frequency and greater spark amplitude. This study confirms the loss of T-tubules as part of the phenotypic change in the failing human myocyte, but it also shows that this is part of a wider spectrum of alterations in surface morphology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

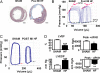

References
-
- Rosengren A, Hauptman P. Women, men and heart failure: A review. Heart Fail Monit. 2008;6:34–40. - PubMed
-
- Lo R, Hsia HH. Ventricular arrhythmias in heart failure patients. Cardiol Clin. 2008;26:381–403. vi. - PubMed
-
- Bardy GH, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005;352:225–237. - PubMed
-
- Mozaffarian D, et al. Prediction of mode of death in heart failure: The Seattle Heart Failure Model. Circulation. 2007;116:392–398. - PubMed
-
- Zipes DP, Wellens HJJ. Sudden cardiac death. Circulation. 1998;98:2334–2351. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

