Imaging of human brain tumor tissue by near-infrared laser coherence tomography
- PMID: 19343270
- PMCID: PMC3085760
- DOI: 10.1007/s00701-009-0248-y
Imaging of human brain tumor tissue by near-infrared laser coherence tomography
Abstract
Introduction: Intraoperative detection of residual tumor remains an important challenge in surgery to treat gliomas. New developments in optical techniques offer non-invasive high-resolution imaging that may integrate well into the workflow of neurosurgical operations. Using an intracranial glioma model, we have recently shown that time domain optical coherence tomography (OCT) allows discrimination of normal brain, diffusely invaded brain tissue, and solid tumor. OCT imaging allowed acquisition of 2D and 3D data arrays for multiplanar analysis of the tumor to brain interface. In this study we have analyzed biopsy specimens of human brain tumors and we present the first feasibility study of intraoperative OCT and post-image acquisition processing for non-invasive imaging of the brain and brain tumor.
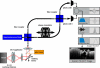
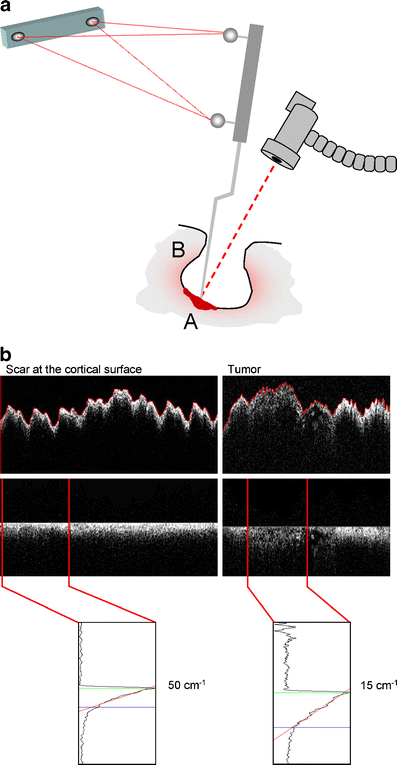
Methods: We used a Sirius 713 Tomograph with a superluminescence diode emitting light at a near infrared central wavelength of 1,310 nm and a coherence length of 15 microm. The light is passed through an optical mono mode fiber to a modified OCT adapter containing a lens system with a working distance of 10 cm and an integrated pilot laser. Navigation-registered tumor biopsies were imaged ex vivo and the intraoperative site of optical tissue analysis was registered by marker acquisition using a neuronavigation system.
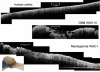
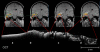
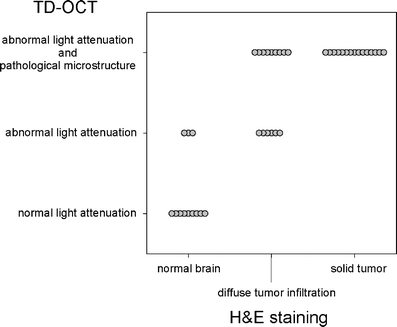
Results: Optical coherence tomography non-contact measurements of brain and brain tumor tissue produced B-scan images of 4 mm in width and 1.5-2.0 mm in depth at an axial and lateral optical resolution of 15 microm. OCT imaging demonstrated a different microstructure and characteristic signal attenuation profiles of tumor versus normal brain. Post-image acquisition processing and automated detection of the tissue to air interface was used to realign A-scans to compensate for image distortions caused by pulse- and respiration-induced movements of the target volume. Realigned images allowed monitoring of intensity changes within the scan line and facilitated selection of areas for the averaging of A-scans and the calculation of attenuation coefficients for specific regions of interest.
Conclusion: This feasibility study has demonstrated that OCT analysis of the tissue microstructure and light attenuation characteristics discriminate normal brain, areas of tumor infiltrated brain, solid tumor, and necrosis. The working distance of the OCT adapter and the A-scan acquisition rate conceptually allows integration of the OCT applicator into the optical path of the operating microscopes. This would allow a continuous analysis of the resection plain, providing optical tomography, thereby adding a third dimension to the microscopic view and information on the light attenuation characteristics of the tissue.
Figures


References
-
- Albert FK, Forsting M, Sartor K, Adams HP, Kunze S. Early postoperative magnetic resonance imaging after resection of malignant glioma: objective evaluation of residual tumor and its influence on regrowth and prognosis. Neurosurgery. 1994;34:45–61. doi: 10.1097/00006123-199401000-00008. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

