Changes in dry state hemoglobin over time do not increase the potential for oxidative DNA damage in dried blood
- PMID: 19352435
- PMCID: PMC2662409
- DOI: 10.1371/journal.pone.0005110
Changes in dry state hemoglobin over time do not increase the potential for oxidative DNA damage in dried blood
Abstract
Background: Hemoglobin (Hb) is the iron-containing oxygen transport protein present in the red blood cells of vertebrates. Ancient DNA and forensic scientists are particularly interested in Hb reactions in the dry state because both regularly encounter aged, dried bloodstains. The DNA in such stains may be oxidatively damaged and, in theory, may be deteriorated by the presence of Hb. To understand the nature of the oxidative systems potentially available to degrade DNA in the presence of dried Hb, we need to determine what molecular species Hb forms over time. These species will determine what type of iron (i.e. Fe(2+)/Fe(3+)/Fe(4+)) is available to participate in further chemical reactions. The availability of "free" iron will affect the ability of the system to undergo Fenton-type reactions which generate the highly reactive hydroxyl radical (OH*). The OH* can directly damage DNA.
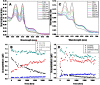
Methodology/principal findings: Oxygenated Hb (oxyHb) converts over time to oxidized Hb (metHb), but this happens more quickly in the dry state than in the hydrated state, as shown by monitoring stabilized oxyHb. In addition, dry state oxyHb converts into at least one other unknown species other than metHb. Although "free" iron was detectable as both Fe(2+) and Fe(3+) in dry and hydrated oxyHb and metHb, the amount of ions detected did not increase over time. There was no evidence that Hb becomes more prone to generating OH* as it ages in either the hydrated or dry states.
Conclusions: The Hb molecule in the dried state undergoes oxidative changes and releases reactive Fe(II) cations. These changes, however, do not appear to increase the ability of Hb to act as a more aggressive Fenton reagent over time. Nevertheless, the presence of Hb in the vicinity of DNA in dried bloodstains creates the opportunity for OH*-induced oxidative damage to the deoxyribose sugar and the DNA nucleobases.
Conflict of interest statement
Figures

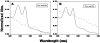




References
-
- Goldstein S, Meyerstein D, Czapski G. The Fenton reagent. Free Radical Biol Med. 1993;15:435–445. - PubMed
-
- Gutteridge JMC. Thiobarbituric acid-reactivity following iron-dependent free-radical damage to amino acids and carbohydrates. FEBS Letters. 1981;128:343–346. - PubMed
-
- Valko M, Morris H, Cronin MT. Metals, toxicity and oxidative stress. Curr Med Chem. 2005;12:1161–1208. - PubMed
-
- Goswami T, Rolfs A, Hediger MA. Iron transport: emerging roles in health and disease. Biochem Cell Biol. 2002;80:679–689. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

