Modulation of Cx46 hemichannels by nitric oxide
- PMID: 19357237
- PMCID: PMC2692414
- DOI: 10.1152/ajpcell.00054.2009
Modulation of Cx46 hemichannels by nitric oxide
Abstract
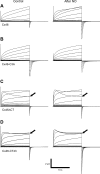
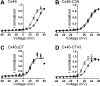
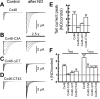
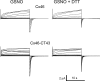
Gap-junction hemichannels are composed of six protein subunits (connexins). Undocked hemichannels contribute to physiological autocrine/paracrine cell signaling, including release of signaling molecules, cell-volume regulation, and glucose uptake. In addition, hemichannels may be pathologically activated by dephosphorylation and cell-membrane depolarization. Such hemichannel opening may induce and/or accelerate cell death. It has been suggested that connexin43 (Cx43) hemichannels are sensitive to redox potential changes and that one or more intracellular cysteines is/are important for this process. Cx46 is expressed in the lens, and its dysfunction induces cataract formation. It contains six cysteines in the extracellular loops, one in the fourth transmembrane helix, and two in the COOH-terminal domain. The latter may be susceptible to oxidation by nitric oxide (NO), which could be involved in cataract formation through cysteine S-nitrosylation. Here we report studies of the effects of the NO donor S-nitrosoglutathione (GSNO) on the electrical properties and fluorescent-dye permeability of wild-type Cx46 and mutant hemichannels expressed in Xenopus laevis oocytes. GSNO enhanced hemichannel voltage sensitivity, increased tail-current amplitude, and changed activation and closing kinetics in Cx46 and Cx46-CT43 (Cx46 mutant in which the COOH terminus was replaced with that of Cx43), but not in Cx46-C3A (Cx46 in which the intracellular and transmembrane helix 4 cysteines were mutated to alanine). We conclude that Cx46 hemichannels are sensitive to NO and that the NO effects are mediated by modification of one or more intracellular cysteines. However, it is unlikely that NO induces cataract formation due to the hemichannel activation, because at normal resting potential, NO had no major effects on Cx46 hemichannel permeability.
Figures
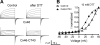

Similar articles
-
Cx46 hemichannel modulation by nitric oxide: Role of the fourth transmembrane helix cysteine and its possible involvement in cataract formation.Nitric Oxide. 2019 May 1;86:54-62. doi: 10.1016/j.niox.2019.02.007. Epub 2019 Feb 21. Nitric Oxide. 2019. PMID: 30797972
-
Functional analysis of hemichannels and gap-junctional channels formed by connexins 43 and 46.Mol Vis. 2010 Jul 15;16:1343-52. Mol Vis. 2010. PMID: 20664797 Free PMC article.
-
Cataract-associated connexin 46 mutation alters its interaction with calmodulin and function of hemichannels.J Biol Chem. 2018 Feb 16;293(7):2573-2585. doi: 10.1074/jbc.RA117.001348. Epub 2018 Jan 3. J Biol Chem. 2018. PMID: 29298900 Free PMC article.
-
Emerging issues of connexin channels: biophysics fills the gap.Q Rev Biophys. 2001 Aug;34(3):325-472. doi: 10.1017/s0033583501003705. Q Rev Biophys. 2001. PMID: 11838236 Review.
-
Regulation of Connexin Gap Junctions and Hemichannels by Calcium and Calcium Binding Protein Calmodulin.Int J Mol Sci. 2020 Nov 2;21(21):8194. doi: 10.3390/ijms21218194. Int J Mol Sci. 2020. PMID: 33147690 Free PMC article. Review.
Cited by
-
Connexin and Pannexin hemichannels are regulated by redox potential.Front Physiol. 2014 Feb 25;5:80. doi: 10.3389/fphys.2014.00080. eCollection 2014. Front Physiol. 2014. PMID: 24611056 Free PMC article. Review.
-
Regulation of gap junction channels and hemichannels by phosphorylation and redox changes: a revision.BMC Cell Biol. 2016 May 24;17 Suppl 1(Suppl 1):11. doi: 10.1186/s12860-016-0099-3. BMC Cell Biol. 2016. PMID: 27229925 Free PMC article. Review.
-
Identification and classification of innexin gene transcripts in the central nervous system of the terrestrial slug Limax valentianus.PLoS One. 2021 Apr 15;16(4):e0244902. doi: 10.1371/journal.pone.0244902. eCollection 2021. PLoS One. 2021. PMID: 33857131 Free PMC article.
-
Replacement of a single cysteine in the fourth transmembrane region of zebrafish pannexin 1 alters hemichannel gating behavior.Exp Brain Res. 2009 Dec;199(3-4):255-64. doi: 10.1007/s00221-009-1957-4. Exp Brain Res. 2009. PMID: 19701745
-
Engram formation in psychiatric disorders.Front Neurosci. 2014 May 28;8:118. doi: 10.3389/fnins.2014.00118. eCollection 2014. Front Neurosci. 2014. PMID: 24904262 Free PMC article. Review.
References
-
- Bao X, Altenberg GA, Reuss L. Mechanism of regulation of the gap junction protein connexin 43 by protein kinase C-mediated phosphorylation. Am J Physiol Cell Physiol 286: C647–C654, 2004. - PubMed
-
- Besancon E, Guo S, Lok J, Tymianski M, Lo EH. Beyond NMDA and AMPA glutamate receptors: emerging mechanisms for ionic imbalance and cell death in stroke. Trends Pharmacol Sci 29: 268–275, 2008. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

