Estimating the location and spatial extent of a covert anthrax release
- PMID: 19360099
- PMCID: PMC2663800
- DOI: 10.1371/journal.pcbi.1000356
Estimating the location and spatial extent of a covert anthrax release
Abstract
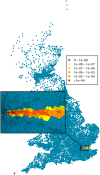
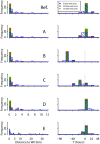
Rapidly identifying the features of a covert release of an agent such as anthrax could help to inform the planning of public health mitigation strategies. Previous studies have sought to estimate the time and size of a bioterror attack based on the symptomatic onset dates of early cases. We extend the scope of these methods by proposing a method for characterizing the time, strength, and also the location of an aerosolized pathogen release. A back-calculation method is developed allowing the characterization of the release based on the data on the first few observed cases of the subsequent outbreak, meteorological data, population densities, and data on population travel patterns. We evaluate this method on small simulated anthrax outbreaks (about 25-35 cases) and show that it could date and localize a release after a few cases have been observed, although misspecifications of the spore dispersion model, or the within-host dynamics model, on which the method relies can bias the estimates. Our method could also provide an estimate of the outbreak's geographical extent and, as a consequence, could help to identify populations at risk and, therefore, requiring prophylactic treatment. Our analysis demonstrates that while estimates based on the first ten or 15 observed cases were more accurate and less sensitive to model misspecifications than those based on five cases, overall mortality is minimized by targeting prophylactic treatment early on the basis of estimates made using data on the first five cases. The method we propose could provide early estimates of the time, strength, and location of an aerosolized anthrax release and the geographical extent of the subsequent outbreak. In addition, estimates of release features could be used to parameterize more detailed models allowing the simulation of control strategies and intervention logistics.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Re-assessment of mitigation strategies for deliberate releases of anthrax using a real-time outbreak characterization tool.Epidemics. 2010 Dec;2(4):189-94. doi: 10.1016/j.epidem.2010.06.001. Epub 2010 Jun 10. Epidemics. 2010. PMID: 21352789
-
The Bayesian aerosol release detector: an algorithm for detecting and characterizing outbreaks caused by an atmospheric release of Bacillus anthracis.Stat Med. 2007 Dec 20;26(29):5225-52. doi: 10.1002/sim.3093. Stat Med. 2007. PMID: 17948918
-
A Bayesian approach for estimating bioterror attacks from patient data.Stat Med. 2011 Jan 30;30(2):101-26. doi: 10.1002/sim.4090. Epub 2010 Oct 20. Stat Med. 2011. PMID: 20963771
-
Preparedness for an anthrax attack.Mol Aspects Med. 2009 Dec;30(6):503-10. doi: 10.1016/j.mam.2009.07.002. Epub 2009 Jul 18. Mol Aspects Med. 2009. PMID: 19619577 Review.
-
[The return of anthrax. From bioterrorism to the zoonotic cluster of Sciacca district].Infez Med. 2010 Jun;18(2):86-90. Infez Med. 2010. PMID: 20610930 Review. Italian.
Cited by
-
Using mixture density networks to emulate a stochastic within-host model of Francisella tularensis infection.PLoS Comput Biol. 2023 Dec 20;19(12):e1011266. doi: 10.1371/journal.pcbi.1011266. eCollection 2023 Dec. PLoS Comput Biol. 2023. PMID: 38117811 Free PMC article.
-
Retracing micro-epidemics of Chagas disease using epicenter regression.PLoS Comput Biol. 2011 Sep;7(9):e1002146. doi: 10.1371/journal.pcbi.1002146. Epub 2011 Sep 15. PLoS Comput Biol. 2011. PMID: 21935346 Free PMC article.
-
Backtracking: Improved methods for identifying the source of a deliberate release of Bacillus anthracis from the temporal and spatial distribution of cases.PLoS Comput Biol. 2024 Sep 6;20(9):e1010817. doi: 10.1371/journal.pcbi.1010817. eCollection 2024 Sep. PLoS Comput Biol. 2024. PMID: 39241092 Free PMC article.
-
Best practice assessment of disease modelling for infectious disease outbreaks.Epidemiol Infect. 2018 Jul;146(10):1207-1215. doi: 10.1017/S095026881800119X. Epub 2018 May 8. Epidemiol Infect. 2018. PMID: 29734964 Free PMC article. Review.
-
A dose and time response Markov model for the in-host dynamics of infection with intracellular bacteria following inhalation: with application to Francisella tularensis.J R Soc Interface. 2014 Jun 6;11(95):20140119. doi: 10.1098/rsif.2014.0119. Print 2014 Jun 6. J R Soc Interface. 2014. PMID: 24671937 Free PMC article.
References
-
- Inglesby TV, O'Toole T, Henderson DA, Bartlett JG, Ascher MS, et al. Anthrax as a biological weapon, 2002: updated recommendations for management. Jama. 2002;287:2236–2252. - PubMed
-
- Brookmeyer R, Blades N. Prevention of inhalational anthrax in the U.S. outbreak. Science. 2002;295:1861. - PubMed
-
- Brookmeyer R, Blades N. Statistical Models and Bioterrorism: Application to the US Anthrax Outbreak. Journal of the American Statistical Association. 2003;98:781–789.
-
- Hogan WR, Cooper GF, Wallstrom GL, Wagner MM, Depinay JM. The Bayesian aerosol release detector: An algorithm for detecting and characterizing outbreaks caused by an atmospheric release of Bacillus anthracis. Stat Med. 2007;26:5225–5252. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

