The Mycobacterium tuberculosis phagosome is a HLA-I processing competent organelle
- PMID: 19360129
- PMCID: PMC2661020
- DOI: 10.1371/journal.ppat.1000374
The Mycobacterium tuberculosis phagosome is a HLA-I processing competent organelle
Abstract
Mycobacterium tuberculosis (Mtb) resides in a long-lived phagosomal compartment that resists maturation. The manner by which Mtb antigens are processed and presented on MHC Class I molecules is poorly understood. Using human dendritic cells and IFN-gamma release by CD8(+) T cell clones, we examined the processing and presentation pathway for two Mtb-derived antigens, each presented by a distinct HLA-I allele (HLA-Ia versus HLA-Ib). Presentation of both antigens is blocked by the retrotranslocation inhibitor exotoxin A. Inhibitor studies demonstrate that, after reaching the cytosol, both antigens require proteasomal degradation and TAP transport, but differ in the requirement for ER-golgi egress and new protein synthesis. Specifically, presentation by HLA-B8 but not HLA-E requires newly synthesized HLA-I and transport through the ER-golgi. Phenotypic analysis of the Mtb phagosome by flow organellometry revealed the presence of Class I and loading accessory molecules, including TAP and PDI. Furthermore, loaded HLA-I:peptide complexes are present within the Mtb phagosome, with a pronounced bias towards HLA-E:peptide complexes. In addition, protein analysis also reveals that HLA-E is enriched within the Mtb phagosome compared to HLA-A2. Together, these data suggest that the phagosome, through acquisition of ER-localized machinery and as a site of HLA-I loading, plays a vital role in the presentation of Mtb-derived antigens, similar to that described for presentation of latex bead-associated antigens. This is, to our knowledge, the first description of this presentation pathway for an intracellular pathogen. Moreover, these data suggest that HLA-E may play a unique role in the presentation of phagosomal antigens.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
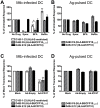


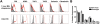
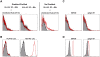
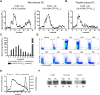
References
-
- Corbett EL, Watt CJ, Walker N, Maher D, Williams BG, et al. The growing burden of tuberculosis: global trends and interactions with the HIV epidemic. Arch Intern Med. 2003;163:1009–1021. - PubMed
-
- Vergne I, Chua J, Singh SB, Deretic V. Cell biology of mycobacterium tuberculosis phagosome. Annu Rev Cell Dev Biol. 2004;20:367–394. - PubMed
-
- Flynn JL, Chan J. Immunology of tuberculosis. Annu Rev Immunol. 2001;19:93–129. - PubMed
-
- Grotzke JE, Lewinsohn DM. Role of CD8+ T lymphocytes in control of Mycobacterium tuberculosis infection. Microbes Infect. 2005;7:776–788. - PubMed
-
- Shen L, Rock KL. Priming of T cells by exogenous antigen cross-presented on MHC class I molecules. Curr Opin Immunol. 2006;18:85–91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

