Liver stem cells and molecular signaling pathways in hepatocellular carcinoma
- PMID: 19360142
- PMCID: PMC2666844
Liver stem cells and molecular signaling pathways in hepatocellular carcinoma
Abstract
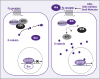
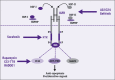
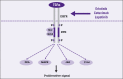
Hepatocellular carcinoma (HCC) is one of the most lethal cancers. Surgical intervention is the only curative option, with only a small fraction of patients being eligible. Conventional chemotherapy and radiotherapy have not been effective in treating this disease, thus leaving patients with an extremely poor prognosis. In viral, alcoholic, and other chronic hepatitis, it has been shown that there is an activation of the progenitor/stem cell population, which has been found to reside in the canals of Hering. In fact, the degree of inflammation and the disease stage have been correlated with the degree of activation. Dysregulation of key regulatory signaling pathways such as transforming growth factor-beta/transforming growth factor-beta receptor (TGF-beta/TBR), insulin-like growth factor/IGF-1 receptor (IGF/IGF-1R), hepatocyte growth factor (HGF/MET), Wnt/beta-catenin/FZD, and transforming growth factor-alpha/epidermal growth factor receptor (TGF-alpha/EGFR) in this progenitor/stem cell population could give rise to HCC. Further understanding of these key signaling pathways and the molecular and genetic alterations associated with HCC could provide major advances in new therapeutic and diagnostic modalities.
Figures



References
-
- Bosch FX, Erdinger L, Ingel F, et al. Primary liver cancer: worldwide incidence and trends. Gastroenterology. 2004;127(suppl 1):S5–S16. - PubMed
-
- Parkin DM, Bray F, Ferlay J, et al. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001;94:153–156. - PubMed
-
- El-Serag HB, Mason AC. Rising incidence of hepatocellular carcinoma in the United States. N Engl J Med. 1999;340:745–750. - PubMed
-
- Roberts LR, Gores GJ. Hepatocellular carcinoma: molecular pathways and new therapeutic targets. Semin Liver Dis. 2005;25:212–225. - PubMed
-
- El-Serag HB. Hepatocellular carcinoma: recent trends in the United States. Gastroenterology. 2004;127(suppl 1):S27–S34. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
