Immune profile of squamous metaplasia development in autoimmune regulator-deficient dry eye
- PMID: 19365590
- PMCID: PMC2660375
Immune profile of squamous metaplasia development in autoimmune regulator-deficient dry eye
Abstract
Purpose: Squamous metaplasia of the ocular surface epithelium in severe Sjögren syndrome (SS) dry eye has been implicated to be associated with chronic engagement of immune-mediated inflammation. While the detailed immunopathological mechanism underlying keratinization of the ocular muco-epithelium in this setting remains unclear, mice deficient in the autoimmune regulator gene (Aire) demonstrate SS-like pathological changes in the exocrine organs and ocular surface including squamous metaplasia. Using this murine model, we sought to determine the specific immune events that predict squamous metaplasia of the cornea in Aire deficiency.
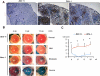
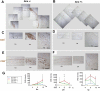
Methods: Lissamine green staining, goblet cell density, and corneal small proline-rich protein 1B (SPRR1B) were compared in Aire-sufficient and -deficient mice at 4, 8, and 16 weeks of age. Corneal, limbal and conjunctival infiltration of CD4(+) and CD8(+) T cells as well as CD11c(+) and MHC class II (I-A(d+)) dendritic cells (DCs) were examined at the same time points. Ordinary least squares regression was used to model SPRR1B's relationship with lissamine green staining, goblet cell density, and immune cell infiltration.
Results: Lissamine green staining was present in Aire-deficient mice by four weeks of age and increased over time. Compared to Aire-sufficient controls, conjunctival goblet cell density (GCD) decreased and corneal SPRR1B increased in Aire-deficient mice with significant differences noted at both 8 and 16 weeks. Immune-mediated CD4(+) T cell infiltration of the conjunctiva and limbus peaked at eight weeks and then decreased. In contrast, corneal T cell infiltration continued to increase over time, reaching a maximum cell number at 16 weeks. CD11c(+) myeloid-derived DCs were found in the conjunctiva and limbus at all time points. As the mice aged, there was a notable increase in corneal CD11c(+) cell counts. Interestingly, the dynamic of activated MHC class II(+) DCs was nearly identical to that of CD4(+) T cells, peaking first in the limbus at eight weeks with maximum infiltration of the cornea by 16 weeks. Regression analysis showed that squamous metaplasia biomarker, SPRR1B, is strongly related to the lissamine green staining of the ocular surface. Corneal infiltration of activated DCs was most prognostic of corneal SPRR1B expression while the presence of precursor DCs, activated DCs, and CD4(+) T cells in the limbus were also significant predictors of SPRR1B.
Conclusions: Aire-deficient mice represent a useful model to study Sjögren-like autoimmune-mediated ocular surface disease. Results of the current study suggest that squamous cell precursor protein, SPRR1B, provides an important readout to evaluate ocular surface damage and specific events related to immune-mediated inflammation. Results also define an appropriate time frame for interventional studies to develop more effective therapies for keratinizing ocular surface disease.
Figures





References
-
- Niederkorn JY, Stern ME, Pflugfelder SC, De Paiva CS, Corrales RM, Gao J, Siemasko K. Desiccating stress induces T cell-mediated Sjogren's Syndrome-like lacrimal keratoconjunctivitis. J Immunol. 2006;176:3950–7. - PubMed
-
- Fox RI, Stern M, Michelson P. Update in Sjogren syndrome. Curr Opin Rheumatol. 2000;12:391–8. - PubMed
-
- Tseng SC. Staging of conjunctival squamous metaplasia by impression cytology. Ophthalmology. 1985;92:728–33. - PubMed
-
- Murube J, Rivas L. Impression cytology on conjunctiva and cornea in dry eye patients establishes a correlation between squamous metaplasia and dry eye clinical severity. Eur J Ophthalmol. 2003;13:115–27. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
