Cetuximab in combination with irinotecan/5-fluorouracil/folinic acid (FOLFIRI) in the initial treatment of metastatic colorectal cancer: a multicentre two-part phase I/II study
- PMID: 19366444
- PMCID: PMC2678147
- DOI: 10.1186/1471-2407-9-112
Cetuximab in combination with irinotecan/5-fluorouracil/folinic acid (FOLFIRI) in the initial treatment of metastatic colorectal cancer: a multicentre two-part phase I/II study
Abstract
Background: This study was designed to investigate the efficacy and safety of the epidermal growth factor receptor (EGFR) inhibitor cetuximab combined with irinotecan, folinic acid (FA) and two different doses of infusional 5-fluorouracil (5-FU) in the first-line treatment of EGFR-detectable metastatic colorectal cancer.
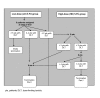
Methods: The 5-FU dose was selected on the basis of dose-limiting toxicities (DLTs) during part I of the study. Patients received cetuximab (400 mg/m2 initial dose and 250 mg/m2/week thereafter) and every 2 weeks irinotecan (180 mg/m2), FA (400 mg/m2) and 5-FU (either low dose [LD], 300 mg/m2 bolus plus 2,000 mg/m2 46-hour infusion, n = 7; or, high-dose [HD], 400 mg/m2 bolus plus 2,400 mg/m2; n = 45).
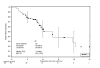
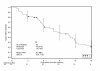
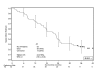
Results: Only two DLTs occurred in the HD group, and HD 5-FU was selected for use in part II. Apart from rash, commonly observed grade 3/4 adverse events such as leucopenia, diarrhoea, vomiting and asthenia occurred within the expected range for FOLFIRI. Among 52 patients, the overall response rate was 48%. Median progression-free survival (PFS) was 8.6 months (counting all reported progressions) and the median overall survival was 22.4 months. Treatment facilitated the resection of initially unresectable metastases in fourteen patients (27%): of these, 10 patients (71%) had no residual tumour after surgery, and these resections hindered the estimation of PFS.
Conclusion: The combination of cetuximab and FOLFIRI was active and well tolerated in this setting. Initially unresectable metastases became resectable in one-quarter of patients, with a high number of complete resections, and these promising results formed the basis for the investigation of FOLFIRI with and without cetuximab in the phase III CRYSTAL trial.
Figures
Similar articles
-
Dose escalating study of cetuximab and 5-FU/folinic acid (FA)/oxaliplatin/irinotecan (FOLFOXIRI) in first line therapy of patients with metastatic colorectal cancer.BMC Cancer. 2014 Jul 19;14:521. doi: 10.1186/1471-2407-14-521. BMC Cancer. 2014. PMID: 25038824 Free PMC article. Clinical Trial.
-
Cetuximab and irinotecan/5-fluorouracil/folinic acid is a safe combination for the first-line treatment of patients with epidermal growth factor receptor expressing metastatic colorectal carcinoma.Ann Oncol. 2006 Mar;17(3):450-6. doi: 10.1093/annonc/mdj084. Epub 2005 Nov 22. Ann Oncol. 2006. PMID: 16303861 Clinical Trial.
-
Efficacy and safety of bevacizumab-based combination regimens in patients with previously untreated metastatic colorectal cancer: final results from a randomised phase II study of bevacizumab plus 5-fluorouracil, leucovorin plus irinotecan versus bevacizumab plus capecitabine plus irinotecan (FNCLCC ACCORD 13/0503 study).Eur J Cancer. 2013 Apr;49(6):1236-45. doi: 10.1016/j.ejca.2012.12.011. Epub 2013 Jan 24. Eur J Cancer. 2013. PMID: 23352604 Clinical Trial.
-
Cetuximab plus XELIRI or XELOX for first-line therapy of metastatic colorectal cancer.Clin Colorectal Cancer. 2008 Mar;7(2):110-7. doi: 10.3816/CCC.2008.n.015. Clin Colorectal Cancer. 2008. PMID: 18501070 Review.
-
Secondary hepatic resection as a therapeutic goal in advanced colorectal cancer.World J Gastroenterol. 2009 Aug 21;15(31):3855-64. doi: 10.3748/wjg.15.3855. World J Gastroenterol. 2009. PMID: 19701965 Free PMC article. Review.
Cited by
-
Cetuximab: a guide to its use in combination with FOLFIRI in the first-line treatment of metastatic colorectal cancer in the USA.Mol Diagn Ther. 2012 Oct;16(5):317-22. doi: 10.1007/s40291-012-0007-2. Mol Diagn Ther. 2012. PMID: 23055389 Review.
-
Synchronous oesophagectomy and hepatic resection for metastatic oesophageal cancer: report of a case.Hippokratia. 2010 Oct;14(4):291-3. Hippokratia. 2010. PMID: 21311643 Free PMC article.
-
Modern therapeutic approaches for hepatic tumors: progress, limitations, and future directions.Discov Oncol. 2025 May 30;16(1):959. doi: 10.1007/s12672-025-02773-z. Discov Oncol. 2025. PMID: 40445470 Free PMC article. Review.
-
Efficacy of the monoclonal antibody EGFR inhibitors for the treatment of metastatic colorectal cancer.Curr Oncol. 2010 Jul;17 Suppl 1(Suppl 1):S3-17. doi: 10.3747/co.v17is1.616. Curr Oncol. 2010. PMID: 20680105 Free PMC article.
-
A molecular sub-cluster of colon cancer cells with low VDR expression is sensitive to chemotherapy, BRAF inhibitors and PI3K-mTOR inhibitors treatment.Aging (Albany NY). 2019 Oct 9;11(19):8587-8603. doi: 10.18632/aging.102349. Epub 2019 Oct 9. Aging (Albany NY). 2019. PMID: 31596728 Free PMC article.
References
-
- Douillard JY, Cunningham D, Roth AD, Navarro M, James RD, Karasek P, Jandik P, Iveson T, Carmichael J, Alakl M. Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet. 2000;355(9209):1041–1047. doi: 10.1016/S0140-6736(00)02034-1. - DOI - PubMed
-
- de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer G. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18(16):2938–2947. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous