Deletion of P2X3 receptors blunts gastro-oesophageal sensation in mice
- PMID: 19368663
- PMCID: PMC2837463
- DOI: 10.1111/j.1365-2982.2009.01292.x
Deletion of P2X3 receptors blunts gastro-oesophageal sensation in mice
Abstract
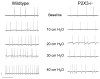
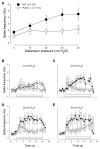
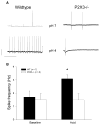
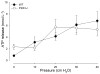
Prior studies have demonstrated P2X receptor expression in the majority of nodose neurons. Immunoreactivity for P2X receptors has also been seen in putative gastric mechanoreceptors, the intraganglionic laminar endings. We therefore hypothesized that deletion of P2X3 receptors will blunt responses to gastric distension in vagal sensory neurons. Using wildtype and P2X3(-/-) mice, we examined responses to purinergic agonists in retrogradely labelled gastric sensory neurons with patch-clamp techniques. Activation of gastro-oesophageal neurons by fluid distension was studied with intracellular electrodes. Distension-evoked ATP release into the gastric lumen was determined with the luciferase assay and intake and gastric emptying of a solid meal was assessed. ATP triggered inward currents in 80% of gastric nodose neurons. In P2X3(-/-) mice, the peak current density was lower compared to controls. Ten of 14 controls but none of 30 neurons from P2X3(-/-) mice responded to alpha,beta-metATP. Gastro-oesophageal sensory neurons of P2X3(-/-) mice showed a blunted response to fluid distension of oesophagus and stomach. This difference was not explained by differences in distension-evoked ATP release, which did not differ between knockout mice and controls. Food intake during a 3-h period was lower in P2X3(-/-) mice. Gastric emptying of a solid meal was slightly faster in knockout mice after 1.5 h, but did not differ between groups at 3 h. Our data support a role of purinergic signalling in gastric vagal afferents. Considering the role of vagal input in sensations of fullness or nausea, P2X receptors may be interesting treatment targets for dyspeptic symptoms.
Figures
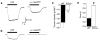

References
-
- Burnstock G. Purinergic nerves. Pharmacol Rev. 1972;24:509–81. - PubMed
-
- Burnstock G. Physiology and pathophysiology of purinergic neurotransmission. Physiol Rev. 2007;87:659–797. - PubMed
-
- Birder LA, Barrick SR, Roppolo JR, et al. Feline interstitial cystitis results in mechanical hypersensitivity and altered ATP release from bladder urothelium. Am J Physiol Renal Physiol. 2003;285:F423–9. - PubMed
-
- Birder LA, Ruan HZ, Chopra B, et al. Alterations in P2X and P2Y purinergic receptor expression in urinary bladder from normal cats and cats with interstitial cystitis. Am J Physiol Renal Physiol. 2004;287:F1084–91. - PubMed
-
- Cockayne DA, Hamilton SG, Zhu QM, et al. Urinary bladder hyporeflexia and reduced pain-related behaviour in P2X3-deficient mice. Nature. 2000;407:1011–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

