Dissecting the functional domains of a nonenveloped virus membrane penetration peptide
- PMID: 19369344
- PMCID: PMC2698515
- DOI: 10.1128/JVI.02299-08
Dissecting the functional domains of a nonenveloped virus membrane penetration peptide
Abstract
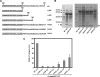
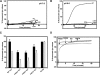
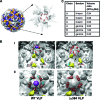
Recent studies have established that several nonenveloped viruses utilize virus-encoded lytic peptides for host membrane disruption. We investigated this mechanism with the "gamma" peptide of the insect virus Flock House virus (FHV). We demonstrate that the C terminus of gamma is essential for membrane disruption in vitro and the rescue of immature virus infectivity in vivo, and the amphipathic N terminus of gamma alone is not sufficient. We also show that deletion of the C-terminal domain disrupts icosahedral ordering of the amphipathic helices of gamma in the virus. Our results have broad implications for understanding membrane lysis during nonenveloped virus entry.
Figures
References
-
- Banerjee, M., and J. E. Johnson. 2008. Activation, exposure and penetration of virally encoded, membrane-active polypeptides during non-enveloped virus entry. Curr. Protein Pept Sci. 916-27. - PubMed
-
- Bong, D. T., C. Steinem, A. Janshoff, J. E. Johnson, and M. Reza Ghadiri. 1999. A highly membrane-active peptide in Flock House virus: implications for the mechanism of nodavirus infection. Chem. Biol. 6473-481. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

