Kainate receptors act as conditional amplifiers of spike transmission at hippocampal mossy fiber synapses
- PMID: 19369569
- PMCID: PMC6665330
- DOI: 10.1523/JNEUROSCI.5807-08.2009
Kainate receptors act as conditional amplifiers of spike transmission at hippocampal mossy fiber synapses
Abstract
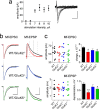
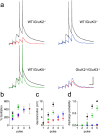
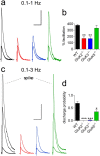
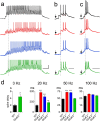
Hippocampal mossy fiber (Mf) synapses are viewed as conditional detonators, assisting CA3 cells in complex network functions. By analyzing mice deficient for GluK2 (GluR6), GluK3 (GluR7) and GluK5 (KA2) genes we show that kainate receptors (KARs) play a crucial role in the control of synaptic integration and spike transmission efficacy at Mf synapses. We dissected out the role of the different KAR functions at Mf synapses and we show that presynaptic and postsynaptic KARs concur to amplify unitary Mf synaptic inputs to trigger spike discharge within a wide range of frequencies (from 1 to 50 Hz). Moreover, KARs strongly favor spike transmission in response to patterns of presynaptic activity mimicking in vivo dentate granule cell activity. By amplifying spike transmission, KARs also facilitate the induction of associative long-term potentiation in CA3. Hence the actions of KARs as amplifiers of spike transmission contribute largely to the "conditional detonator" function of Mf synapses and are likely important for spatial information processing.
Figures
Similar articles
-
Selective block of postsynaptic kainate receptors reveals their function at hippocampal mossy fiber synapses.Cereb Cortex. 2013 Feb;23(2):323-31. doi: 10.1093/cercor/bhs022. Epub 2012 Feb 17. Cereb Cortex. 2013. PMID: 22345355
-
Distinct subunits in heteromeric kainate receptors mediate ionotropic and metabotropic function at hippocampal mossy fiber synapses.J Neurosci. 2005 Dec 14;25(50):11710-8. doi: 10.1523/JNEUROSCI.4041-05.2005. J Neurosci. 2005. PMID: 16354929 Free PMC article.
-
Loss of kainate receptor-mediated heterosynaptic facilitation of mossy-fiber synapses in KA2-/- mice.J Neurosci. 2003 Jan 15;23(2):422-9. doi: 10.1523/JNEUROSCI.23-02-00422.2003. J Neurosci. 2003. PMID: 12533602 Free PMC article.
-
Presynaptic kainate receptors at hippocampal mossy fiber synapses.Proc Natl Acad Sci U S A. 2001 Sep 25;98(20):11003-8. doi: 10.1073/pnas.191351498. Proc Natl Acad Sci U S A. 2001. PMID: 11572960 Free PMC article. Review.
-
GLUK1 receptor antagonists and hippocampal mossy fiber function.Int Rev Neurobiol. 2009;85:13-27. doi: 10.1016/S0074-7742(09)85002-2. Int Rev Neurobiol. 2009. PMID: 19607958 Review.
Cited by
-
Homeostatic control of synaptic transmission by distinct glutamate receptors.Neuron. 2013 May 22;78(4):687-99. doi: 10.1016/j.neuron.2013.02.031. Neuron. 2013. PMID: 23719165 Free PMC article.
-
Net(o) excitement for kainate receptors.Nat Neurosci. 2011 Jun 27;14(7):808-10. doi: 10.1038/nn.2864. Nat Neurosci. 2011. PMID: 21709676 No abstract available.
-
Glutamate receptor ion channels: structure, regulation, and function.Pharmacol Rev. 2010 Sep;62(3):405-96. doi: 10.1124/pr.109.002451. Pharmacol Rev. 2010. PMID: 20716669 Free PMC article. Review.
-
Reversal of hippocampal neuronal maturation by serotonergic antidepressants.Proc Natl Acad Sci U S A. 2010 May 4;107(18):8434-9. doi: 10.1073/pnas.0912690107. Epub 2010 Apr 19. Proc Natl Acad Sci U S A. 2010. PMID: 20404165 Free PMC article.
-
Synaptic activation of kainate receptors gates presynaptic CB(1) signaling at GABAergic synapses.Nat Neurosci. 2010 Feb;13(2):197-204. doi: 10.1038/nn.2481. Epub 2010 Jan 17. Nat Neurosci. 2010. PMID: 20081851
References
-
- Bischofberger J, Engel D, Frotscher M, Jonas P. Timing and efficacy of transmitter release at mossy fiber synapses in the hippocampal network. Pflugers Arch. 2006;453:361–372. - PubMed
-
- Castillo PE, Malenka RC, Nicoll RA. Kainate receptors mediate a slow postsynaptic current in hippocampal CA3 neurons. Nature. 1997;388:182–186. - PubMed
-
- Contractor A, Swanson G, Heinemann SF. Kainate receptors are involved in short- and long-term plasticity at mossy fiber synapses in the hippocampus. Neuron. 2001;29:209–216. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
