GPIHBP1 and lipolysis: an update
- PMID: 19369870
- PMCID: PMC2810420
- DOI: 10.1097/MOL.0b013e32832ac026
GPIHBP1 and lipolysis: an update
Abstract
Purpose of review: This review will provide an update on the structure of GPIHBP1, a 28-kDa glycosylphosphatidylinositol-anchored glycoprotein, and its role in the lipolytic processing of triglyceride-rich lipoproteins.
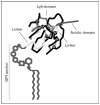
Recent findings: Gpihbp1 knockout mice on a chow diet have milky plasma and plasma triglyceride levels of more than 3000 mg/dl. GPIHBP1 is located on the luminal surface of endothelial cells in tissues where lipolysis occurs: heart, skeletal muscle, and adipose tissue. The pattern of lipoprotein lipase (LPL) release into the plasma after an intravenous injection of heparin is abnormal in Gpihbp1-deficient mice, suggesting that GPIHBP1 plays a direct role in binding LPL within the tissues of mice. Transfection of CHO cells with a GPIHBP1 expression vector confers on cells the ability to bind both LPL and chylomicrons. Two regions of GPIHBP1 are required for the binding of LPL - an amino-terminal acidic domain and the cysteine-rich Ly6 domain. GPIHBP1 expression in mice changes with fasting and refeeding and is regulated in part by peroxisome proliferator-activated receptor-gamma.
Summary: GPIHBP1, an endothelial cell-surface glycoprotein, binds LPL and is required for the lipolytic processing of triglyceride-rich lipoproteins.
Conflict of interest statement
There was no conflict of interest.
Figures


Similar articles
-
Glycosylphosphatidylinositol-anchored high-density lipoprotein-binding protein 1 plays a critical role in the lipolytic processing of chylomicrons.Cell Metab. 2007 Apr;5(4):279-91. doi: 10.1016/j.cmet.2007.02.002. Cell Metab. 2007. PMID: 17403372 Free PMC article.
-
The expression of GPIHBP1, an endothelial cell binding site for lipoprotein lipase and chylomicrons, is induced by peroxisome proliferator-activated receptor-gamma.Mol Endocrinol. 2008 Nov;22(11):2496-504. doi: 10.1210/me.2008-0146. Epub 2008 Sep 11. Mol Endocrinol. 2008. PMID: 18787041 Free PMC article.
-
GPIHBP1, a GPI-anchored protein required for the lipolytic processing of triglyceride-rich lipoproteins.J Lipid Res. 2009 Apr;50 Suppl(Suppl):S57-62. doi: 10.1194/jlr.R800030-JLR200. Epub 2008 Oct 14. J Lipid Res. 2009. PMID: 18854402 Free PMC article. Review.
-
Highly conserved cysteines within the Ly6 domain of GPIHBP1 are crucial for the binding of lipoprotein lipase.J Biol Chem. 2009 Oct 30;284(44):30240-7. doi: 10.1074/jbc.M109.046391. Epub 2009 Sep 2. J Biol Chem. 2009. PMID: 19726683 Free PMC article.
-
GPIHBP1: an endothelial cell molecule important for the lipolytic processing of chylomicrons.Curr Opin Lipidol. 2007 Aug;18(4):389-96. doi: 10.1097/MOL.0b013e3281527914. Curr Opin Lipidol. 2007. PMID: 17620854 Free PMC article. Review.
Cited by
-
GPIHBP1 and the processing of triglyceride-rich lipoproteins.Clin Lipidol. 2010 Aug 1;5(4):575-582. doi: 10.2217/clp.10.43. Clin Lipidol. 2010. PMID: 20953295 Free PMC article.
-
Stabilizing lipoprotein lipase.J Lipid Res. 2009 Dec;50(12):2335-6. doi: 10.1194/jlr.E000703. Epub 2009 Aug 6. J Lipid Res. 2009. PMID: 19661257 Free PMC article. No abstract available.
-
Functional validation of GPIHBP1 and identification of a functional mutation in GPIHBP1 for milk fat traits in dairy cattle.Sci Rep. 2017 Aug 17;7(1):8546. doi: 10.1038/s41598-017-08668-6. Sci Rep. 2017. PMID: 28819221 Free PMC article.
-
Identification and quantitative mRNA analysis of a novel splice variant of GPIHBP1 in dairy cattle.J Anim Sci Biotechnol. 2014 Nov 5;5(1):50. doi: 10.1186/2049-1891-5-50. eCollection 2014. J Anim Sci Biotechnol. 2014. PMID: 25810903 Free PMC article.
-
Emerging Role of Lymphocyte Antigen-6 Family of Genes in Cancer and Immune Cells.Front Immunol. 2019 Apr 24;10:819. doi: 10.3389/fimmu.2019.00819. eCollection 2019. Front Immunol. 2019. PMID: 31068932 Free PMC article. Review.
References
-
- Brunzell JD, Deeb SS. Familial lipoprotein lipase deficiency, apo C-II deficiency, and hepatic lipase deficiency. In: Scriver CR, Beaudet AL, Sly WS, et al., editors. The metabolic and molecular bases of inherited disease. 8. New York: McGraw-Hill; 2001. pp. 2789–2816.
-
- Havel RJ, Kane JP. Introduction: structure and metabolism of plasma lipoproteins. In: Scriver CR, Beaudet AL, Sly WS, et al., editors. The metabolic and molecular bases of inherited disease. 8. New York: McGraw-Hill; 2001. pp. 2705–2716.
-
- Tan MH, Sata T, Havel RJ. The significance of lipoprotein lipase in rat skeletal muscles. J Lipid Res. 1977;18:363–370. - PubMed
-
- Ioka RX, Kang M-J, Kamiyama S, et al. Expression cloning and characterization of a novel glycosylphosphatidylinositol-anchored high density lipoprotein-binding protein, GPI-HBP1. J Biol Chem. 2003;278:7344–7349. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

