Molecular dissection of Alzheimer's disease neuropathology by depletion of serum amyloid P component
- PMID: 19372378
- PMCID: PMC2669789
- DOI: 10.1073/pnas.0902640106
Molecular dissection of Alzheimer's disease neuropathology by depletion of serum amyloid P component
Abstract
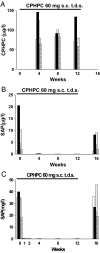
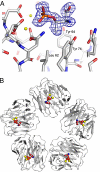
New therapeutic approaches in Alzheimer's disease are urgently needed. The normal plasma protein, serum amyloid P component (SAP), is always present in cerebrospinal fluid (CSF) and in the pathognomonic lesions of Alzheimer's disease, cerebrovascular and intracerebral Abeta amyloid plaques and neurofibrillary tangles, as a result of its binding to amyloid fibrils and to paired helical filaments, respectively. SAP itself may also be directly neurocytotoxic. Here, in this unique study in Alzheimer's disease of the bis(d-proline) compound, (R)-1-[6-[(R)-2-carboxy-pyrrolidin-1-yl]-6-oxo-hexanoyl]pyrrolidine-2-carboxylic acid (CPHPC), we observed depletion of circulating SAP and also remarkable, almost complete, disappearance of SAP from the CSF. We demonstrate that SAP depletion in vivo is caused by CPHPC cross-linking pairs of SAP molecules in solution to form complexes that are immediately cleared from the plasma. We have also solved the structure of SAP complexed with phosphothreonine, its likely ligand on hyperphosphorylated tau protein. These results support further clinical study of SAP depletion in Alzheimer's disease and potentially other neurodegenerative diseases.
Conflict of interest statement
Conflict of interest: M.B.P. is the inventor on patents related to SAP and CPHPC that are owned by Pentraxin Therapeutics Ltd., a University College London spinout company in which he, P.N.H., and S.P.W. have shares, which owns CPHPC.
Figures

References
-
- Pepys MB, et al. Amyloid P component. A critical review. Amyloid: Int J Exp Clin Invest. 1997;4:274–295.
-
- Hawkins PN, et al. Concentration of serum amyloid P component in the CSF as a possible marker of cerebral amyloid deposits in Alzheimer's disease. Biochem Biophys Res Commun. 1994;201:722–726. - PubMed
-
- Coria F, et al. Isolation and characterization of amyloid P component from Alzheimer's disease and other types of cerebral amyloidosis. Lab Invest. 1988;58:454–458. - PubMed
-
- Duong T, Pommier EC, Scheibel AB. Immunodetection of the amyloid P component in Alzheimer's disease. Acta Neuropathol. 1989;78:429–437. - PubMed
-
- Kalaria RN, Grahovac I. Serum amyloid P immunoreactivity in hippocampal tangles, plaques and vessels: implications for leakage across the blood-brain barrier in Alzheimer's disease. Brain Res. 1990;516:349–353. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

