Curcumin circumvents chemoresistance in vitro and potentiates the effect of thalidomide and bortezomib against human multiple myeloma in nude mice model
- PMID: 19372569
- PMCID: PMC2694943
- DOI: 10.1158/1535-7163.MCT-08-0905
Curcumin circumvents chemoresistance in vitro and potentiates the effect of thalidomide and bortezomib against human multiple myeloma in nude mice model
Erratum in
- Mol Cancer Ther. 2009 May;8(5):1398
Abstract
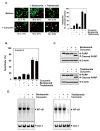
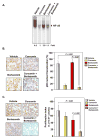
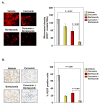
Curcumin (diferuloylmethane), a yellow pigment in turmeric, has been shown to inhibit the activation of nuclear factor-kappaB (NF-kappaB), a transcription factor closely linked to chemoresistance in multiple myeloma cells. Whether curcumin can overcome chemoresistance and enhance the activity of thalidomide and bortezomib, used to treat patients with multiple myeloma, was investigated in vitro and in xenograft model in nude mice. Our results show that curcumin inhibited the proliferation of human multiple myeloma cells regardless of their sensitivity to dexamethasone, doxorubicin, or melphalan. Curcumin also potentiated the apoptotic effects of thalidomide and bortezomib by down-regulating the constitutive activation of NF-kappaB and Akt, and this correlated with the suppression of NF-kappaB-regulated gene products, including cyclin D1, Bcl-xL, Bcl-2, TRAF1, cIAP-1, XIAP, survivin, and vascular endothelial growth factor. Furthermore, in a nude mice model, we found that curcumin potentiated the antitumor effects of bortezomib (P<0.001, vehicle versus bortezomib+curcumin; P<0.001, bortezomib versus bortezomib+curcumin), and this correlated with suppression of Ki-67 (P<0.001 versus control), CD31 (P<0.001 versus vehicle), and vascular endothelial growth factor (P<0.001 versus vehicle) expression. Collectively, our results suggest that curcumin overcomes chemoresistance and sensitizes multiple myeloma cells to thalidomide and bortezomib by down-regulating NF-kappaB and NF-kappaB-regulated gene products.
Conflict of interest statement
Figures



Similar articles
-
Thymoquinone overcomes chemoresistance and enhances the anticancer effects of bortezomib through abrogation of NF-κB regulated gene products in multiple myeloma xenograft mouse model.Oncotarget. 2014 Feb 15;5(3):634-48. doi: 10.18632/oncotarget.1596. Oncotarget. 2014. PMID: 24504138 Free PMC article.
-
Resveratrol inhibits proliferation, induces apoptosis, and overcomes chemoresistance through down-regulation of STAT3 and nuclear factor-kappaB-regulated antiapoptotic and cell survival gene products in human multiple myeloma cells.Blood. 2007 Mar 15;109(6):2293-302. doi: 10.1182/blood-2006-02-003988. Epub 2006 Dec 12. Blood. 2007. PMID: 17164350
-
Curcumin potentiates antitumor activity of gemcitabine in an orthotopic model of pancreatic cancer through suppression of proliferation, angiogenesis, and inhibition of nuclear factor-kappaB-regulated gene products.Cancer Res. 2007 Apr 15;67(8):3853-61. doi: 10.1158/0008-5472.CAN-06-4257. Cancer Res. 2007. PMID: 17440100
-
Current status of bortezomib in the treatment of multiple myeloma.Curr Hematol Malig Rep. 2007 May;2(2):128-37. doi: 10.1007/s11899-007-0018-y. Curr Hematol Malig Rep. 2007. PMID: 20425361 Review.
-
The role of nuclear factor-kappaB in the biology and treatment of multiple myeloma.Semin Oncol. 2001 Dec;28(6):626-33. doi: 10.1016/s0093-7754(01)90036-3. Semin Oncol. 2001. PMID: 11740821 Review.
Cited by
-
Curcumin enhances the lung cancer chemopreventive efficacy of phospho-sulindac by improving its pharmacokinetics.Int J Oncol. 2013 Sep;43(3):895-902. doi: 10.3892/ijo.2013.1995. Epub 2013 Jun 27. Int J Oncol. 2013. PMID: 23807084 Free PMC article.
-
The nontoxic natural compound Curcumin exerts anti-proliferative, anti-migratory, and anti-invasive properties against malignant gliomas.BMC Cancer. 2010 Sep 14;10:491. doi: 10.1186/1471-2407-10-491. BMC Cancer. 2010. PMID: 20840775 Free PMC article.
-
Curcumin enhances cytotoxic effects of bortezomib in human multiple myeloma H929 cells: potential roles of NF-κB/JNK.Int J Mol Sci. 2012;13(4):4831-4838. doi: 10.3390/ijms13044831. Epub 2012 Apr 16. Int J Mol Sci. 2012. PMID: 22606012 Free PMC article.
-
Role of Natural Products in Modulating Histone Deacetylases in Cancer.Molecules. 2019 Mar 16;24(6):1047. doi: 10.3390/molecules24061047. Molecules. 2019. PMID: 30884859 Free PMC article. Review.
-
Active Components of Traditional Chinese Medicinal Material for Multiple Myeloma: Current Evidence and Future Directions.Front Pharmacol. 2022 Jan 27;13:818179. doi: 10.3389/fphar.2022.818179. eCollection 2022. Front Pharmacol. 2022. PMID: 35153791 Free PMC article. Review.
References
-
- Ries LG, Alves MC, Berzin F. Asymmetric activation of temporalis, masseter, and sternocleidomastoid muscles in temporomandibular disorder patients. Cranio. 2008;26:59–64. - PubMed
-
- Bharti AC, Donato N, Singh S, Aggarwal BB. Curcumin (diferuloylmethane) down-regulates the constitutive activation of nuclear factor-kappa B and IkappaBalpha kinase in human multiple myeloma cells, leading to suppression of proliferation and induction of apoptosis. Blood. 2003;101:1053–62. - PubMed
-
- Bharti AC, Shishodia S, Reuben JM, et al. Nuclear factor-kappaB and STAT3 are constitutively active in CD138+ cells derived from multiple myeloma patients, and suppression of these transcription factors leads to apoptosis. Blood. 2004;103:3175–84. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

