The ubiquitin ligase Siah2 and the hypoxia response
- PMID: 19372575
- PMCID: PMC2860273
- DOI: 10.1158/1541-7786.MCR-08-0458
The ubiquitin ligase Siah2 and the hypoxia response
Abstract
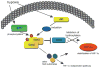
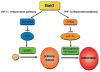
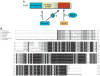
Growing evidence indicates that ubiquitin ligases play a critical role in the hypoxia response. Among them, Siah2, a RING finger ligase, is an important regulator of pathways activated under hypoxia. Siah2 regulates prolyl hydroxylases PHD3 and 1 under oxygen concentration of 2% to 5%, thereby allowing accumulation of hypoxia-inducible factor (HIF)-1alpha, a master regulator of the hypoxia response within the range of physiological normoxic to mild hypoxic conditions. Growing evidence also indicates an important function for Siah2 in tumor development and progression based on pancreatic cancer, mammary tumor, and melanoma mouse models. This review summarizes our current understanding of Siah2 regulation and function with emphasis on hypoxia and tumorigenesis.
Conflict of interest statement
No potential conflicts of interest were disclosed.
Figures
References
-
- Wenger RH, Stiehl DP, Camenisch G. Integration of oxygen signaling at the consensus HRE. Sci STKE. 2005;2005:re12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

