Naturally-existing CD4(+)CD25(+)Foxp3(+) regulatory T cells are required for tolerance to experimental autoimmune thyroiditis induced by either exogenous or endogenous autoantigen
- PMID: 19375891
- PMCID: PMC2706097
- DOI: 10.1016/j.jaut.2009.03.010
Naturally-existing CD4(+)CD25(+)Foxp3(+) regulatory T cells are required for tolerance to experimental autoimmune thyroiditis induced by either exogenous or endogenous autoantigen
Abstract
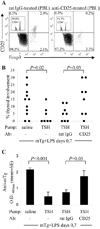
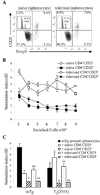
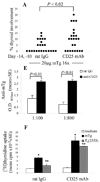
Murine experimental autoimmune thyroiditis (EAT) is a model for Hashimoto's thyroiditis, an organ-specific autoimmune disease characterized by mononuclear cell infiltration and destruction of the thyroid gland. Susceptibility to EAT is MHC-linked, and influenced by CD4(+)CD25(+)Foxp3(+) regulatory T cells (Tregs). Treg depletion enables thyroiditis induction with mouse thyroglobulin (mTg) in traditionally-resistant mice and mTg-induced, Treg-mediated tolerance protects against EAT induction in genetically-susceptible mice. Here, we demonstrate the existence of naturally-existing CD4(+)CD25(+)Foxp3(+) Tregs (nTregs) influencing thyroiditis development in naive susceptible mice and that induction of thyroiditis in these mice involves overcoming peripheral homeostatic immune suppression by nTregs. Additionally we demonstrate that nTregs are required for induction of antigen-specific tolerance, indicating that induced EAT tolerance is a result of activation of naturally-existing nTregs rather than de novo generation of induced Tregs (iTregs). Examination of several potential costimulatory molecules previously described as involved in peripheral activation of Tregs demonstrates a critical role indeed for CTLA-4 in the activation of nTregs leading to development of EAT tolerance and providing a mechanism for mTg-induced Treg activation during tolerance induction. Together, these data reinforce the important role of Tregs in mediating self-tolerance, and illuminate a potential mechanism for their therapeutic expansion in induced tolerance.
Figures



Similar articles
-
Autoimmune thyroiditis: a model uniquely suited to probe regulatory T cell function.J Autoimmun. 2009 Nov-Dec;33(3-4):239-46. doi: 10.1016/j.jaut.2009.09.004. Epub 2009 Oct 12. J Autoimmun. 2009. PMID: 19822405 Free PMC article. Review.
-
Enhanced autoimmunity associated with induction of tumor immunity in thyroiditis-susceptible mice.Thyroid. 2013 Dec;23(12):1590-9. doi: 10.1089/thy.2013.0064. Epub 2013 Sep 11. Thyroid. 2013. PMID: 23777580 Free PMC article.
-
Depletion of CD4+CD25+ regulatory T cells exacerbates sodium iodide-induced experimental autoimmune thyroiditis in human leucocyte antigen DR3 (DRB1*0301) transgenic class II-knock-out non-obese diabetic mice.Clin Exp Immunol. 2007 Mar;147(3):547-54. doi: 10.1111/j.1365-2249.2006.03303.x. Clin Exp Immunol. 2007. PMID: 17302906 Free PMC article.
-
H2A- and H2E-derived CD4+CD25+ regulatory T cells: a potential role in reciprocal inhibition by class II genes in autoimmune thyroiditis.J Immunol. 2005 Mar 1;174(5):3111-6. doi: 10.4049/jimmunol.174.5.3111. J Immunol. 2005. PMID: 15728527
-
Tolerance to autoimmune thyroiditis: (CD4+)CD25+ regulatory T cells influence susceptibility but do not supersede MHC class II restriction.Front Biosci. 2006 May 1;11:1234-43. doi: 10.2741/1876. Front Biosci. 2006. PMID: 16368509 Review.
Cited by
-
Opportunistic autoimmune disorders: from immunotherapy to immune dysregulation.Ann N Y Acad Sci. 2010 Jan;1183:222-36. doi: 10.1111/j.1749-6632.2009.05138.x. Ann N Y Acad Sci. 2010. PMID: 20146718 Free PMC article. Review.
-
Immunotoxicity from checkpoint inhibitor therapy: clinical features and underlying mechanisms.Immunology. 2020 Feb;159(2):167-177. doi: 10.1111/imm.13141. Epub 2019 Nov 19. Immunology. 2020. PMID: 31646612 Free PMC article. Review.
-
Current understanding of CTLA-4: from mechanism to autoimmune diseases.Front Immunol. 2023 Jul 11;14:1198365. doi: 10.3389/fimmu.2023.1198365. eCollection 2023. Front Immunol. 2023. PMID: 37497212 Free PMC article. Review.
-
Efficacy of HLA-DRB1∗03:01 and H2E transgenic mouse strains to correlate pathogenic thyroglobulin epitopes for autoimmune thyroiditis.J Autoimmun. 2011 Sep;37(2):63-70. doi: 10.1016/j.jaut.2011.05.010. Epub 2011 Jun 17. J Autoimmun. 2011. PMID: 21683551 Free PMC article.
-
Thymic deletion and regulatory T cells prevent antimyeloperoxidase GN.J Am Soc Nephrol. 2013 Mar;24(4):573-85. doi: 10.1681/ASN.2012090898. Epub 2013 Feb 7. J Am Soc Nephrol. 2013. PMID: 23393320 Free PMC article.
References
-
- Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor α-chains (CD25): breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 1995;155:1151–1164. - PubMed
-
- Taguchi O, Takahashi T. Administration of anti-interleukin-2 receptor α antibody in vivo induces localized autoimmune disease. Eur. J. Immunol. 1996;26:1608–1612. - PubMed
-
- McHugh RS, Shevach EM. Cutting edge: Depletion of CD4+CD25+ regulatory T cells is necessary, but not sufficient, for induction of organ-specific autoimmune disease. J. Immunol. 2002;168:5979–5983. - PubMed
-
- Morris GP, Yan Y, David CS, Kong YM. H2A- and H2E–derived CD4+CD25+ regulatory T cells: a potential role in reciprocal inhibition by class II genes in autoimmune thyroiditis. J. Immunol. 2005;174:3111–3116. - PubMed
-
- Mottet C, Uhlig HH, Powrie F. Cutting edge: cure of colitis by CD4+CD25+ regulatory T cells. J. Immunol. 2003;170:3939–3943. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

