Parallel processing in the corticogeniculate pathway of the macaque monkey
- PMID: 19376073
- PMCID: PMC2789995
- DOI: 10.1016/j.neuron.2009.02.024
Parallel processing in the corticogeniculate pathway of the macaque monkey
Abstract
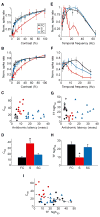
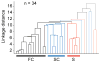
Although corticothalamic feedback is ubiquitous across species and modalities, its role in sensory processing is unclear. This study provides a detailed description of the visual physiology of corticogeniculate neurons in the primate. Using electrical stimulation to identify corticogeniculate neurons, we distinguish three groups of neurons with response properties that closely resemble those of neurons in the magnocellular, parvocellular, and koniocellular layers of their target structure, the lateral geniculate nucleus (LGN) of the thalamus. Our results indicate that corticogeniculate feedback in the primate is stream specific, and provide strong evidence in support of the view that corticothalamic feedback can influence the transmission of sensory information from the thalamus to the cortex in a stream-selective manner.
Figures




References
-
- Ahmed B, Anderson JC, Douglas RJ, Martin KA, Nelson JC. Polyneuronal innervation of spiny stellate neurons in cat visual cortex. J Comp Neurol. 1994;341:39–49. - PubMed
-
- Albrecht DG, Hamilton DB. Striate cortex of monkey and cat: contrast response function. J Neurophys. 1982;48:217–237. - PubMed
-
- Alitto HJ, Usrey WM. Corticothalamic feedback and sensory processing. Cur Op Neurosci. 2003;13:1–6. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

