Intrinsic impaired proplatelet formation and microtubule coil assembly of megakaryocytes in a mouse model of Bernard-Soulier syndrome
- PMID: 19377075
- PMCID: PMC2688571
- DOI: 10.3324/haematol.2008.001032
Intrinsic impaired proplatelet formation and microtubule coil assembly of megakaryocytes in a mouse model of Bernard-Soulier syndrome
Abstract
Background: Giant platelets and thrombocytopenia are invariable defects in the Bernard-Soulier syndrome caused by deficiency of the GPIb-V-IX complex, a receptor for von Willebrand factor supporting platelet adhesion to the damaged arterial wall. Various properties of this receptor may be considered potential determinants of the macrothrombocytopenia.
Design and methods: To explore the underlying mechanisms of the disease, megakaryopoiesis was studied in a mouse model deficient in GPIbbeta. Megakaryocytes were initially characterized in situ in the bone marrow of adult mice, after which their capacity to differentiate into proplatelet-bearing cells was evaluated in cultured fetal liver cells.
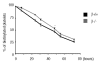
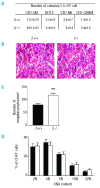
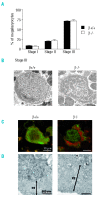
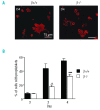
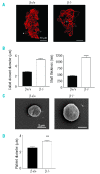
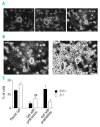
Results: The number of megakaryocyte progenitors, their differentiation and progressive maturation into distinct classes and their level of endoreplication were normal in GPIbbeta(-/-) bone marrow. However, the more mature cells exhibited ultrastructural anomalies with a thicker peripheral zone and a less well developed demarcation membrane system. GPIbbeta(-/-) megakaryocytes could be differentiated in culture from Lin(-) fetal liver cells in normal amounts but the proportion of cells able to extend proplatelets was decreased by 41%. Moreover, the GPIbbeta(-/-) cells extending proplatelets displayed an abnormal morphology characterized by fewer pseudopodial extensions with thicker shaft sections and an increased diameter of the terminal coiled elements. GPIbbeta(-/-) released platelets were larger but retained a typical discoid shape. Proplatelet formation was similarly affected in bone marrow explants from adult mice examined by videomicroscopy. The marginal microtubular ring contained twice as many tubulin fibers in GPIbbeta(-/-) proplatelet buds in cultured and circulating platelets.
Conclusions: Altogether, these findings point to a role of the GPIb-V-IX complex intrinsic to megakaryocytes at the stage of proplatelet formation and suggest a functional link with the underlying microtubular cytoskeleton in platelet biogenesis.
Figures

Comment in
-
The regulation of proplatelet production.Haematologica. 2009 Jun;94(6):756-9. doi: 10.3324/haematol.2009.006577. Haematologica. 2009. PMID: 19483152 Free PMC article. No abstract available.
References
-
- Bernard J, Soulier JP. Sem Hop. 1948;24:3217–23. [Not Available] - PubMed
-
- Nurden AT, Didry D, Rosa JP. Molecular defects of platelets in Bernard-Soulier syndrome. Blood Cells. 1983;9:333–58. - PubMed
-
- Berndt MC, Du XP, Booth WJ. Ristocetin-dependent reconstitution of binding of von Willebrand factor to purified human platelet membrane glycoprotein Ib-IX complex. Biochemistry. 1988;27:633–40. - PubMed
-
- Italiano JE, Jr, Shivdasani RA. Megakaryocytes and beyond: the birth of platelets. J Thromb Haemost. 2003;1:1174–82. - PubMed
-
- Schulze H, Shivdasani RA. Mechanisms of thrombopoiesis. J Thromb Haemost. 2005;3:1717–24. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

