Dissecting the urokinase activation pathway using urokinase-activated anthrax toxin
- PMID: 19377974
- PMCID: PMC3393601
- DOI: 10.1007/978-1-60327-003-8_10
Dissecting the urokinase activation pathway using urokinase-activated anthrax toxin
Abstract
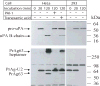
Anthrax toxin is a three-part toxin secreted by Bacillus anthracis, consisting of protective antigen (PrAg), edema factor (EF), and lethal factor (LF). To intoxicate host mammalian cells, PrAg, the cell-binding moiety of the toxin, binds to cells and is then proteolytically activated by furin on the cell surface, resulting in the active heptameric form of PrAg. This heptamer serves as a protein-conducting channel that translocates EF and LF, the two enzymatic moieties of the toxin, into the cytosol of the cells where they exert cytotoxic effects. The anthrax toxin delivery system has been well characterized. The amino-terminal PrAg-binding domain of LF (residues 1-254, LFn) is sufficient to allow translocation of fused "passenger" polypeptides, such as the ADP-ribosylation domain of Pseudomonas exotoxin A, to the cytosol of the cells in a PrAg-dependent process. The protease specificity of the anthrax toxin delivery system can also be reengineered by replacing the furin cleavage target sequence of PrAg with other protease substrate sequences. PrAg-U2 is such a PrAg variant, one that is selectively activated by urokinase plasminogen activator (uPA). The uPA-dependent proteolytic activation of PrAg-U2 on the cell surface is readily detected by western blotting analysis of cell lysates in vitro, or cell or animal death in vivo. Here, we describe the use of PrAg-U2 as a molecular reporter tool to test the controversial question of what components are required for uPAR-mediated cell surface pro-uPA activation. The results demonstrate that both uPAR and plasminogen play critical roles in pro-uPA activation both in vitro and in vivo.
Figures


References
-
- Leppla SH. Bacillus anthracis toxins. In: Alouf JE, Popoff MR, editors. The Comprehensive Sourcebook of Bacterial Protein Toxins. Academic Press; Burlington, MA: 2006. pp. 323–347.
-
- Bradley KA, Mogridge J, Mourez M, Collier RJ, Young JA. Identification of the cellular receptor for anthrax toxin. Nature. 2001;414:225–229. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

