Kidney dendritic cell activation is required for progression of renal disease in a mouse model of glomerular injury
- PMID: 19381017
- PMCID: PMC2673875
- DOI: 10.1172/JCI38399
Kidney dendritic cell activation is required for progression of renal disease in a mouse model of glomerular injury
Erratum in
- J Clin Invest. 2009 Jul;119(7):2114
Abstract
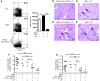
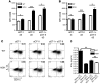
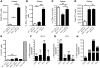
The progression of kidney disease to renal failure correlates with infiltration of mononuclear immune cells into the tubulointerstitium. These infiltrates contain macrophages, DCs, and T cells, but the role of each cell type in disease progression is unclear. To investigate the underlying immune mechanisms, we generated transgenic mice that selectively expressed the model antigens ovalbumin and hen egg lysozyme in glomerular podocytes (NOH mice). Coinjection of ovalbumin-specific transgenic CD8+ CTLs and CD4+ Th cells into NOH mice resulted in periglomerular mononuclear infiltrates and inflammation of parietal epithelial cells, similar to lesions frequently observed in human chronic glomerulonephritis. Repetitive T cell injections aggravated infiltration and caused progression to structural and functional kidney damage after 4 weeks. Mechanistic analysis revealed that DCs in renal lymph nodes constitutively cross-presented ovalbumin and activated CTLs. These CTLs released further ovalbumin for CTL activation in the lymph nodes and for simultaneous presentation to Th cells by distinct DC subsets residing in the kidney tubulointerstitium. Crosstalk between tubulointerstitial DCs and Th cells resulted in intrarenal cytokine and chemokine production and in recruitment of more CTLs, monocyte-derived DCs, and macrophages. The importance of DCs was established by the fact that DC depletion rapidly resolved established kidney immunopathology. These findings demonstrate that glomerular antigen-specific CTLs and Th cells can jointly induce renal immunopathology and identify kidney DCs as a mechanistic link between glomerular injury and the progression of kidney disease.
Figures

Comment in
-
New roles revealed for T cells and DCs in glomerulonephritis.J Clin Invest. 2009 May;119(5):1074-6. doi: 10.1172/jci39071. J Clin Invest. 2009. PMID: 19422093 Free PMC article.
Similar articles
-
T cell cross-talk with kidney dendritic cells in glomerulonephritis.J Mol Med (Berl). 2010 Jan;88(1):19-26. doi: 10.1007/s00109-009-0541-5. Epub 2009 Oct 2. J Mol Med (Berl). 2010. PMID: 19798477 Review.
-
New roles revealed for T cells and DCs in glomerulonephritis.J Clin Invest. 2009 May;119(5):1074-6. doi: 10.1172/jci39071. J Clin Invest. 2009. PMID: 19422093 Free PMC article.
-
Antigen presentation by dendritic cells in renal lymph nodes is linked to systemic and local injury to the kidney.Kidney Int. 2005 Sep;68(3):1096-108. doi: 10.1111/j.1523-1755.2005.00502.x. Kidney Int. 2005. PMID: 16105040
-
Active CD4+ helper T cells directly stimulate CD8+ cytotoxic T lymphocyte responses in wild-type and MHC II gene knockout C57BL/6 mice and transgenic RIP-mOVA mice expressing islet beta-cell ovalbumin antigen leading to diabetes.Autoimmunity. 2008 Nov;41(7):501-11. doi: 10.1080/08916930802069256. Autoimmunity. 2008. PMID: 18855194
-
Kidney dendritic cells: fundamental biology and functional roles in health and disease.Nat Rev Nephrol. 2020 Jul;16(7):391-407. doi: 10.1038/s41581-020-0272-y. Epub 2020 May 5. Nat Rev Nephrol. 2020. PMID: 32372062 Review.
Cited by
-
Regulatory T cells use programmed death 1 ligands to directly suppress autoreactive B cells in vivo.Proc Natl Acad Sci U S A. 2012 Jun 26;109(26):10468-73. doi: 10.1073/pnas.1201131109. Epub 2012 Jun 11. Proc Natl Acad Sci U S A. 2012. PMID: 22689978 Free PMC article.
-
Renal dendritic cells ameliorate nephrotoxic acute kidney injury.J Am Soc Nephrol. 2010 Jan;21(1):53-63. doi: 10.1681/ASN.2009040407. Epub 2009 Oct 29. J Am Soc Nephrol. 2010. PMID: 19875815 Free PMC article.
-
T cell cross-talk with kidney dendritic cells in glomerulonephritis.J Mol Med (Berl). 2010 Jan;88(1):19-26. doi: 10.1007/s00109-009-0541-5. Epub 2009 Oct 2. J Mol Med (Berl). 2010. PMID: 19798477 Review.
-
Cross-priming in health and disease.Nat Rev Immunol. 2010 Jun;10(6):403-14. doi: 10.1038/nri2780. Nat Rev Immunol. 2010. PMID: 20498667 Review.
-
Local renal autoantibody production in lupus nephritis.J Am Soc Nephrol. 2011 Feb;22(2):296-305. doi: 10.1681/ASN.2010050515. Epub 2010 Nov 18. J Am Soc Nephrol. 2011. PMID: 21088295 Free PMC article.
References
-
- Bohle A., Strutz F., Muller G.A. On the pathogenesis of chronic renal failure in primary glomerulopathies: a view from the interstitium. Exp. Nephrol. 1994;2:205–210. - PubMed
-
- Nikolic-Paterson D.J., Atkins R.C. The role of macrophages in glomerulonephritis. Nephrol. Dial. Transplant. 2001;16(Suppl. 5):3–7. - PubMed
-
- Markovic-Lipkovski J., Muller C.A., Risler T., Bohle A., Muller G.A. Association of glomerular and interstitial mononuclear leukocytes with different forms of glomerulonephritis. Nephrol. Dial. Transplant. 1990;5:10–17. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

