Heterogeneity of Kir4.1 channel expression in glia revealed by mouse transgenesis
- PMID: 19382212
- PMCID: PMC2764821
- DOI: 10.1002/glia.20882
Heterogeneity of Kir4.1 channel expression in glia revealed by mouse transgenesis
Abstract
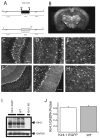
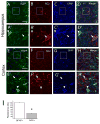
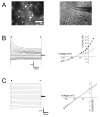
The weakly inwardly rectifying K(+) channel Kir4.1 is found in many glial cells including astrocytes. However, questions remain regarding the relative contribution of Kir4.1 to the resting K(+) conductance of mature astrocytes in situ. We employed a bacterial artificial chromosome transgenic approach in mice to visualize Kir4.1 expression in vivo. These mice (Kir4.1-EGFP) express enhanced green fluorescent protein (EGFP) under the transcriptional control of the Kir4.1 promoter. The brains of adult Kir4.1-EGFP transgenic mice showed co-expression of EGFP and Kir4.1 in astrocytes. In addition, weaker expression of EGFP was detected in NG2+ glial cells when compared with EGFP expression in GFAP+ glial cells. Whole-cell voltage clamp recordings of EGFP+ glial cells in the CA1 area of the adult mouse hippocampus indicated astrocytes displaying properties consistent with both the "passive" and "complex" subpopulations. EGFP+ cells with bright fluorescence had the linear current-voltage (I-V) relationships and extensive gap junctional coupling characteristic of passive astrocytes. However, EGFP+ glia with weaker fluorescence displayed properties associated with complex astrocytes including nonlinear I-V relationships and lack of intercellular gap junctional coupling. Pharmacological blockade of inward currents implied that Kir4.1 channels constitute the dominant resting K(+) conductance in both glial cell types and are more highly expressed in passive astrocytes. These results suggest differential expression of Kir4.1 in glia and that this channel likely underlies the resting K(+) conductance in passive and complex astrocytes.
Figures



References
-
- Barbarese E, Carson JH, Braun PE. Accumulation of four myelin basic proteins in mouse during development. J Neurochem. 1978:779–782. - PubMed
-
- Connors NC, Adams ME, Froehner SC, Kofuji P. The potassium channel Kir4.1 associates with the dystrophin glycoprotein complex via alpha-syntrophin in glia. J Biol Chem. 2004;279:28387–92. - PubMed
-
- Doupnik CA, Davidson N, Lester HA. The inward rectifier potassium channel family. Curr Opin Neurobiol. 1995;5:268–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

