Hereditary ovarian carcinoma: heterogeneity, molecular genetics, pathology, and management
- PMID: 19383374
- PMCID: PMC2778287
- DOI: 10.1016/j.molonc.2009.02.004
Hereditary ovarian carcinoma: heterogeneity, molecular genetics, pathology, and management
Abstract
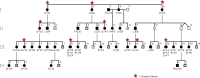
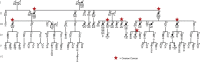
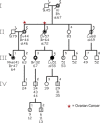
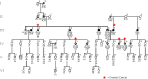
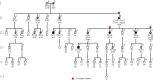
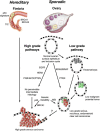
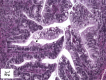
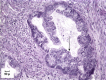
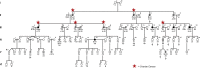
Hereditary ovarian cancer accounts for at least 5% of the estimated 22,000 new cases of this disease during 2009. During this same time, over 15,000 will die from malignancy ascribed to ovarian origin. The bulk of these hereditary cases fits the hereditary breast-ovarian cancer syndrome, while virtually all of the remainder will be consonant with the Lynch syndrome, disorders which are autosomal dominantly inherited. Advances in molecular genetics have led to the identification of BRCA1 and BRCA2 gene mutations which predispose to the hereditary breast-ovarian cancer syndrome, and mutations in mismatch repair genes, the most common of which are MSH2 and MLH1, which predispose to Lynch syndrome. These discoveries enable relatively certain diagnosis, limited only by their variable penetrance, so that identification of mutation carriers through a comprehensive cancer family history might be possible. This paper reviews the subject of hereditary ovarian cancer, with particular attention to its molecular genetic basis, its pathology, and its phenotypic/genotypic heterogeneity.
Figures




References
-
- Aaltonen, L.A. , Peltomaki, P. , Leach, F.S. , Sistonen, P. , Pylkkanen, L. , Mecklin, J.-P. , 1993. Clues to the pathogenesis of familial colorectal cancer. Science. 260, 812–816. - PubMed
-
- Aaltonen, L.A. , Salovaara, R. , Kristo, P. , Canzian, F. , Hemminki, A. , Peltomäki, P. , 1998. Incidence of hereditary nonpolyposis colorectal cancer and the feasibility of molecular screening for the disease. N. Engl. J. Med.. 338, 1481–1487. - PubMed
-
- Aarnio, M. , Sankila, R. , Pukkala, E. , Salovaara, R. , Aaltonen, L.A. , de la Chapelle, A. , Peltomäki, P. , Mecklin, J.-P. , Järvinen, H.J. , 1999. Cancer risk in mutation carriers of DNA-mismatch-repair genes. Int. J. Cancer. 81, 214–218. - PubMed
-
- Agoff, S.N. , Mendelin, J.E. , Grieco, V.S. , Garcia, R.L. , 2002. Unexpected gynecologic neoplasm in patients with proven or suspected BRCA-1 or -2 mutations: implications for gross examination, cytology, and clinical follow-up. Am. J. Surg. Pathol.. 26, 171–178. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

