A highly reproducible rotenone model of Parkinson's disease
- PMID: 19385059
- PMCID: PMC2757935
- DOI: 10.1016/j.nbd.2009.01.016
A highly reproducible rotenone model of Parkinson's disease
Abstract
The systemic rotenone model of Parkinson's disease (PD) accurately replicates many aspects of the pathology of human PD and has provided insights into the pathogenesis of PD. The major limitation of the rotenone model has been its variability, both in terms of the percentage of animals that develop a clear-cut nigrostriatal lesion and the extent of that lesion. The goal here was to develop an improved and highly reproducible rotenone model of PD. In these studies, male Lewis rats in three age groups (3, 7 or 12-14 months) were administered rotenone (2.75 or 3.0 mg/kg/day) in a specialized vehicle by daily intraperitoneal injection. All rotenone-treated animals developed bradykinesia, postural instability, and/or rigidity, which were reversed by apomorphine, consistent with a lesion of the nigrostriatal dopamine system. Animals were sacrificed when the PD phenotype became debilitating. Rotenone treatment caused a 45% loss of tyrosine hydroxylase-positive substantia nigra neurons and a commensurate loss of striatal dopamine. Additionally, in rotenone-treated animals, alpha-synuclein and poly-ubiquitin positive aggregates were observed in dopamine neurons of the substantia nigra. In summary, this version of the rotenone model is highly reproducible and may provide an excellent tool to test new neuroprotective strategies.
Figures

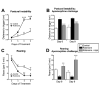




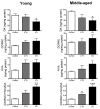
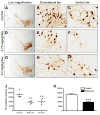
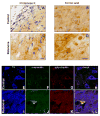
References
-
- Alam M, Schmidt WJ. Rotenone destroys dopaminergic neurons and induces parkinsonian symptoms in rats. Behav Brain Res. 2002;136:317–24. - PubMed
-
- Alam M, Schmidt WJ. L-DOPA reverses the hypokinetic behaviour and rigidity in rotenone-treated rats. Behav Brain Res. 2004;153:439–46. - PubMed
-
- Betarbet R, Canet-Aviles RM, Sherer TB, Mastroberardino PG, McLendon C, Kim JH, Lund S, Na HM, Taylor G, Bence NF, Kopito R, Seo BB, Yagi T, Yagi A, Klinefelter G, Cookson MR, Greenamyre JT. Intersecting pathways to neurodegeneration in Parkinson's disease: effects of the pesticide rotenone on DJ-1, alpha-synuclein, and the ubiquitin-proteasome system. Neurobiol Dis. 2006;22:404–20. - PubMed
-
- Betarbet R, Sherer TB, MacKenzie G, Garcia-Osuna M, Panov AV, Greenamyre JT. Chronic systemic pesticide exposure reproduces features of Parkinson's disease. Nat Neurosci. 2000;3:1301–6. - PubMed
-
- Biehlmaier O, Alam M, Schmidt WJ. A rat model of Parkinsonism shows depletion of dopamine in the retina. Neurochem Int. 2007;50:189–95. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
