Structural mass spectrometry of the alpha beta-tubulin dimer supports a revised model of microtubule assembly
- PMID: 19388626
- PMCID: PMC4129956
- DOI: 10.1021/bi900200q
Structural mass spectrometry of the alpha beta-tubulin dimer supports a revised model of microtubule assembly
Erratum in
- Biochemistry. 2009 Oct 13;48(40):9704
Abstract
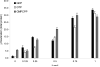
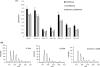
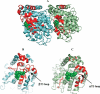
The molecular basis of microtubule lattice instability derives from the hydrolysis of GTP to GDP in the lattice-bound state of alphabeta-tubulin. While this has been appreciated for many years, there is ongoing debate over the molecular basis of this instability and the possible role of altered nucleotide occupancy in the induction of a conformational change in tubulin. The debate has organized around seemingly contradictory models. The allosteric model invokes nucleotide-dependent states of curvature in the free tubulin dimer, such that hydrolysis leads to pronounced bending and thus disruption of the lattice. The more recent lattice model describes a predominant role for the lattice in straightening free dimers that are curved regardless of their nucleotide state. In this model, lattice-bound GTP-tubulin provides the necessary force to straighten an incoming dimer. Interestingly, there is evidence for both models. The enduring nature of this debate stems from a lack of high-resolution data on the free dimer. In this study, we have prepared alphabeta-tubulin samples at high dilution and characterized the nature of nucleotide-induced conformational stability using bottom-up hydrogen/deuterium exchange mass spectrometry (H/DX-MS) coupled with isothermal urea denaturation experiments. These experiments were accompanied by molecular dynamics simulations of the free dimer. We demonstrate an intermediate state unique to GDP-tubulin, suggestive of the curved colchicine-stabilized structure at the intradimer interface but show that intradimer flexibility is an important property of the free dimer regardless of nucleotide occupancy. Our results indicate that the assembly properties of the free dimer may be better described on the basis of this flexibility. A blended model of assembly emerges in which free-dimer allosteric effects retain importance, in an assembly process dominated by lattice-induced effects.
Figures

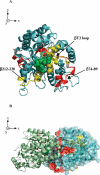
Similar articles
-
Intrinsic bending and structural rearrangement of tubulin dimer: molecular dynamics simulations and coarse-grained analysis.Biophys J. 2008 Sep;95(5):2487-99. doi: 10.1529/biophysj.108.129072. Epub 2008 May 30. Biophys J. 2008. PMID: 18515385 Free PMC article.
-
Microtubule assembly governed by tubulin allosteric gain in flexibility and lattice induced fit.Elife. 2018 Apr 13;7:e34353. doi: 10.7554/eLife.34353. Elife. 2018. PMID: 29652248 Free PMC article.
-
The free energy profile of tubulin straight-bent conformational changes, with implications for microtubule assembly and drug discovery.PLoS Comput Biol. 2014 Feb 6;10(2):e1003464. doi: 10.1371/journal.pcbi.1003464. eCollection 2014 Feb. PLoS Comput Biol. 2014. PMID: 24516374 Free PMC article.
-
Tubulin rings: which way do they curve?Curr Opin Struct Biol. 2003 Apr;13(2):256-61. doi: 10.1016/s0959-440x(03)00029-0. Curr Opin Struct Biol. 2003. PMID: 12727521 Review.
-
How tubulin subunits are lost from the shortening ends of microtubules.J Struct Biol. 1997 Mar;118(2):107-18. doi: 10.1006/jsbi.1997.3844. J Struct Biol. 1997. PMID: 9126637 Review.
Cited by
-
Statistical mechanics provides novel insights into microtubule stability and mechanism of shrinkage.PLoS Comput Biol. 2015 Feb 18;11(2):e1004099. doi: 10.1371/journal.pcbi.1004099. eCollection 2015 Feb. PLoS Comput Biol. 2015. PMID: 25692909 Free PMC article.
-
Mechanical properties of tubulin intra- and inter-dimer interfaces and their implications for microtubule dynamic instability.PLoS Comput Biol. 2019 Aug 30;15(8):e1007327. doi: 10.1371/journal.pcbi.1007327. eCollection 2019 Aug. PLoS Comput Biol. 2019. PMID: 31469822 Free PMC article.
-
Modeling the effects of drug binding on the dynamic instability of microtubules.Phys Biol. 2011 Oct;8(5):056004. doi: 10.1088/1478-3975/8/5/056004. Epub 2011 Aug 12. Phys Biol. 2011. PMID: 21836336 Free PMC article.
-
Designing and Testing of Novel Taxanes to Probe the Highly Complex Mechanisms by Which Taxanes Bind to Microtubules and Cause Cytotoxicity to Cancer Cells.PLoS One. 2015 Jun 8;10(6):e0129168. doi: 10.1371/journal.pone.0129168. eCollection 2015. PLoS One. 2015. PMID: 26052950 Free PMC article.
-
GTP-dependent formation of straight tubulin oligomers leads to microtubule nucleation.J Cell Biol. 2021 Apr 5;220(4):e202007033. doi: 10.1083/jcb.202007033. J Cell Biol. 2021. PMID: 33544140 Free PMC article.
References
-
- Desai A, Mitchison TJ. Microtubule polymerization dynamics. Annu. Rev. Cell Dev. Biol. 1997;13:83–117. - PubMed
-
- Menendez M, Rivas G, Diaz JF, Andreu JM. Control of the structural stability of the tubulin dimer by one high affinity bound magnesium ion at nucleotide N-site. J. Biol. Chem. 1998;273:167–176. - PubMed
-
- Lowe J, Li H, Downing KH, Nogales E. Refined structure of alpha beta-tubulin at 3.5Å resolution. J. Mol. Biol. 2001;313:1045–1057. - PubMed
-
- Carlier MF, Pantaloni D. Kinetic analysis of guanosine 5′-triphosphate hydrolysis associated with tubulin polymerization. Biochemistry. 1981;20:1918–1924. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

