Twisted gastrulation limits apoptosis in the distal region of the mandibular arch in mice
- PMID: 19389368
- PMCID: PMC2851169
- DOI: 10.1016/j.ydbio.2008.12.041
Twisted gastrulation limits apoptosis in the distal region of the mandibular arch in mice
Abstract
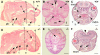
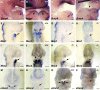
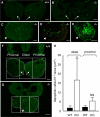
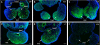
The mandibular arch (BA1) is critical for craniofacial development. The distal region of BA1, which gives rise to most of the mandible, is dependent upon an optimal level of bone morphogenetic protein (BMP) signaling. BMP activity is modulated in the extracellular space by BMP-binding proteins such as Twisted gastrulation (TWSG1). Twsg1(-/-) mice have a spectrum of craniofacial phenotypes, including mandibular defects that range from micrognathia to agnathia. At E9.5, the distal region of the mutant BA1 was prematurely and variably fused with loss of distal markers eHand and Msx1. Expression of proximal markers Fgf8 and Barx1 was expanded across the fused BA1. The expression of Bmp4 and Msx2 was preserved in the distal region, but shifted ventrally. While wild type embryos showed a gradient of BMP signaling with higher activity in the distal region of BA1, this gradient was disrupted and shifted ventrally in the mutants. Thus, loss of TWSG1 results in disruption of the BMP4 gradient at the level of signaling activity as well as mRNA expression. Altered distribution of BMP signaling leads to a shift in gene expression and increase in apoptosis. The extent of apoptosis may account for the variable degree of mandibular defects in Twsg1 mutants.
Figures




Similar articles
-
The BMP antagonists Chordin and Noggin have essential but redundant roles in mouse mandibular outgrowth.Dev Biol. 2001 Dec 15;240(2):457-73. doi: 10.1006/dbio.2001.0479. Dev Biol. 2001. PMID: 11784076
-
Threshold-specific requirements for Bmp4 in mandibular development.Dev Biol. 2005 Jul 15;283(2):282-93. doi: 10.1016/j.ydbio.2005.04.019. Dev Biol. 2005. PMID: 15936012
-
YY1 activates Msx2 gene independent of bone morphogenetic protein signaling.Nucleic Acids Res. 2002 Mar 1;30(5):1213-23. doi: 10.1093/nar/30.5.1213. Nucleic Acids Res. 2002. PMID: 11861914 Free PMC article.
-
Multifaceted Functions of TWSG1: From Embryogenesis to Cancer Development.Int J Mol Sci. 2022 Oct 22;23(21):12755. doi: 10.3390/ijms232112755. Int J Mol Sci. 2022. PMID: 36361543 Free PMC article. Review.
-
Reassessing the Dlx code: the genetic regulation of branchial arch skeletal pattern and development.J Anat. 2005 Nov;207(5):501-61. doi: 10.1111/j.1469-7580.2005.00487.x. J Anat. 2005. PMID: 16313391 Free PMC article. Review.
Cited by
-
Enhanced osteoclastogenesis causes osteopenia in twisted gastrulation-deficient mice through increased BMP signaling.J Bone Miner Res. 2009 Nov;24(11):1917-26. doi: 10.1359/jbmr.090507. J Bone Miner Res. 2009. PMID: 19419314 Free PMC article.
-
Bone morphogenetic protein 2 signaling in osteoclasts is negatively regulated by the BMP antagonist, twisted gastrulation.J Cell Biochem. 2011 Mar;112(3):793-803. doi: 10.1002/jcb.23003. J Cell Biochem. 2011. PMID: 21328453 Free PMC article.
-
GWAS of dental caries patterns in the permanent dentition.J Dent Res. 2013 Jan;92(1):38-44. doi: 10.1177/0022034512463579. Epub 2012 Oct 11. J Dent Res. 2013. PMID: 23064961 Free PMC article.
-
Autosomal dominant mesomandibular fibro-osseous dysplasia: a self-resolving inherited fibro-osseous lesion of the jaws.Front Physiol. 2012 Dec 6;3:458. doi: 10.3389/fphys.2012.00458. eCollection 2012. Front Physiol. 2012. PMID: 23230423 Free PMC article.
-
Loss of Neogenin alters branchial arch development and leads to craniofacial skeletal defects.Front Cell Dev Biol. 2024 Feb 9;12:1256465. doi: 10.3389/fcell.2024.1256465. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 38404688 Free PMC article.
References
-
- Ben-Zvi D, Shilo BZ, Fainsod A, Barkai N. Scaling of the BMP activation gradient in Xenopus embryos. Nature. 2008;453:1205–11. - PubMed
-
- Bollenbach T, Pantazis P, Kicheva A, Bokel C, Gonzalez-Gaitan M, Julicher F. Precision of the Dpp gradient. Development. 2008;135:1137–46. - PubMed
-
- Cerny R, Lwigale P, Ericsson R, Meulemans D, Epperlein HH, Bronner-Fraser M. Developmental origins and evolution of jaws: new interpretation of “maxillary” and “mandibular”. Dev Biol. 2004;276:225–36. - PubMed
-
- Chai Y, Jiang X, Ito Y, Bringas P, Jr., Han J, Rowitch DH, Soriano P, McMahon AP, Sucov HM. Fate of the mammalian cranial neural crest during tooth and mandibular morphogenesis. Development. 2000;127:1671–9. - PubMed
-
- Chai Y, Sasano Y, Bringas P, Jr., Mayo M, Kaartinen V, Heisterkamp N, Groffen J, Slavkin H, Shuler C. Characterization of the fate of midline epithelial cells during the fusion of mandibular prominences in vivo. Dev Dyn. 1997;208:526–35. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

