Ascl1 and Neurog2 form novel complexes and regulate Delta-like3 (Dll3) expression in the neural tube
- PMID: 19389376
- PMCID: PMC2698949
- DOI: 10.1016/j.ydbio.2009.01.007
Ascl1 and Neurog2 form novel complexes and regulate Delta-like3 (Dll3) expression in the neural tube
Abstract
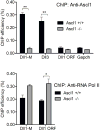
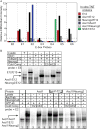
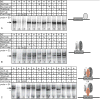
Delta-like 3 (Dll3) is a Delta family member expressed broadly in the developing nervous system as neural progenitor cells initiate differentiation. A proximal promoter sequence for Dll3 is conserved across multiple species and is sufficient to direct GFP expression in a Dll3-like pattern in the neural tube of transgenic mice. This promoter contains multiple E-boxes, the consensus binding site for bHLH factors. Dll3 expression and the activity of the Dll3-promoter in the dorsal neural tube depends on the basic helix-loop-helix (bHLH) transcription factors Ascl1 (Mash1) and Neurog2 (Ngn2). Mutations in each E-box identified in the Dll3-promoter allowed distinct enhancer or repressor properties to be assigned to each site individually or in combination. In addition, each E-box has distinct characteristics relative to binding of bHLH factors Ascl1, Neurog1, and Neurog2. Surprisingly, novel Ascl1 containing DNA binding complexes are identified that interact with specific E-box sites within the Dll3-promoter in vitro. These complexes include Ascl1/Ascl1 homodimers and Ascl1/Neurog2 heterodimers, complexes that in some cases require additional undefined factors for efficient DNA binding. Thus, a complex interplay of E-box binding proteins spatially and temporally regulate Dll3 levels during neural tube development.
Figures




References
-
- Anand G, Yin X, Shahidi AK, Grove L, Prochownik EV. Novel regulation of the helix-loop-helix protein Id1 by S5a, a subunit of the 26 S proteasome. J Biol Chem. 1997;272:19140–51. - PubMed
-
- Bertrand N, Castro DS, Guillemot F. Proneural genes and the specification of neural cell types. Nat Rev Neuroscience. 2002;3:517–530. - PubMed
-
- Bettenhausen B, Hrabe de Angelis M, Simon D, Guenet J-L, Gossler A. Transient and restricted expression during mouse embryogenesis of Dll1, a murine gene closely related to Drosophila Delta. Development. 1995;121:2407–2418. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

