On-bead screening of combinatorial libraries: reduction of nonspecific binding by decreasing surface ligand density
- PMID: 19397369
- PMCID: PMC2765537
- DOI: 10.1021/cc9000168
On-bead screening of combinatorial libraries: reduction of nonspecific binding by decreasing surface ligand density
Abstract
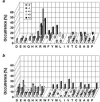
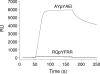
On-bead screening of one-bead-one-compound (OBOC) libraries provides a powerful method for the rapid identification of active compounds against molecular or cellular targets. However, on-bead screening is susceptible to interference from nonspecific binding, which results in biased screening data and false positives. In this work, we have found that a major source of nonspecific binding is derived from the high ligand loading on the library beads, which permits a macromolecular target (e.g., a protein) to simultaneously interact with multiple ligands on the bead surface. To circumvent this problem, we have synthesized a phosphotyrosyl (pY)-containing peptide library on spatially segregated TentaGel microbeads, which feature a 10-fold reduced peptide loading on the bead surface but a normal peptide loading in the bead interior. The library was screened against a panel of 10 Src homology 2 (SH2) domains including those of Csk and Fyn kinases and adaptor protein SLAP, and the specific recognition motif(s) was successfully identified for each of the domains. In contrast, when the SH2 domains were screened against a control library that contained unaltered (high) ligand loading at the bead surface, six of them exhibited varying degrees of sequence biases, ranging from minor perturbation in the relative abundance of different sequences to the exclusive selection of false positive sequences that have no measurable affinity to the target protein. These results indicate that reduction of the ligand loading on the bead surface represents a simple, effective strategy to largely eliminate the interference from nonspecific binding, while preserving sufficient amounts of materials in the bead interior for compound identification. This finding should further expand the utility of OBOC libraries in biomedical research.
Figures
Similar articles
-
Defining SH2 domain and PTP specificity by screening combinatorial peptide libraries.Methods. 2007 Jul;42(3):207-19. doi: 10.1016/j.ymeth.2007.02.010. Methods. 2007. PMID: 17532507 Free PMC article.
-
Determination of the binding specificity of the SH2 domains of protein tyrosine phosphatase SHP-1 through the screening of a combinatorial phosphotyrosyl peptide library.Biochemistry. 2000 Oct 31;39(43):13251-60. doi: 10.1021/bi0014397. Biochemistry. 2000. PMID: 11052678
-
Functional diversity of Csk, Chk, and Src SH2 domains due to a single residue variation.J Biol Chem. 2005 Jul 8;280(27):25780-7. doi: 10.1074/jbc.M504022200. Epub 2005 May 12. J Biol Chem. 2005. PMID: 15890649
-
From combinatorial chemistry to cancer-targeting peptides.Mol Pharm. 2007 Sep-Oct;4(5):631-51. doi: 10.1021/mp700073y. Epub 2007 Sep 20. Mol Pharm. 2007. PMID: 17880166 Review.
-
Applications of one-bead one-compound combinatorial libraries and chemical microarrays in signal transduction research.Acc Chem Res. 2003 Jun;36(6):370-7. doi: 10.1021/ar0201299. Acc Chem Res. 2003. PMID: 12809522 Review.
Cited by
-
Profiling the substrate specificity of protein kinases by on-bead screening of peptide libraries.Biochemistry. 2013 Aug 20;52(33):5645-55. doi: 10.1021/bi4008947. Epub 2013 Jul 24. Biochemistry. 2013. PMID: 23848432 Free PMC article.
-
Discovery of a Direct Ras Inhibitor by Screening a Combinatorial Library of Cell-Permeable Bicyclic Peptides.ACS Comb Sci. 2016 Jan 11;18(1):75-85. doi: 10.1021/acscombsci.5b00164. Epub 2015 Dec 23. ACS Comb Sci. 2016. PMID: 26645887 Free PMC article.
-
Multiplexed Competitive Screening of One-Bead-One-Component Combinatorial Libraries Using a ClonePix 2 Colony Sorter.Int J Mol Sci. 2019 Oct 16;20(20):5119. doi: 10.3390/ijms20205119. Int J Mol Sci. 2019. PMID: 31623061 Free PMC article.
-
hνSABR: Photochemical Dose-Response Bead Screening in Droplets.Anal Chem. 2016 Mar 1;88(5):2904-11. doi: 10.1021/acs.analchem.5b04811. Epub 2016 Feb 13. Anal Chem. 2016. PMID: 26815064 Free PMC article.
-
Utility of redundant combinatorial libraries in distinguishing high and low quality screening hits.ACS Comb Sci. 2014 Jun 9;16(6):259-70. doi: 10.1021/co500030f. Epub 2014 Apr 21. ACS Comb Sci. 2014. PMID: 24749624 Free PMC article.
References
-
-
For examples see: Chen JK, Lane WS, Brauer AW, Tanaka A, Schreiber SL. J. Am. Chem. Soc. 1993;115:12591–12592.Muller K, Gombert FO, Manning U, Grossmuller F, Graff P, Zaegel H, Zuber JF, Freuler F, Tschopp C, Baumann G. J. Biol. Chem. 1996;271:16500–16505.Smith HK, Bradley M. J. Comb. Chem. 1999;1:326–332.Alluri PG, Reddy MM, Bachhawat-Sikder K, Olivos HJ, Kodadek T. J. Am. Chem. Soc. 2003;125:13995–14004.Sweeney MC, Wavreille A-S, Park J, Butchar J, Tridandapani S, Pei D. Biochemistry. 2005;44:14932–14947.Peng L, Liu RW, Marik J, Wang X-B, Takada Y, Lam KS. Nature Chem. Biol. 2006;2:381–389.Zhang Y, Zhou S, Wavreille A-S, DeWille J, Pei D. J. Comb. Chem. 2008;10:247–255.
-
-
-
For examples see: Wu J, Ma QN, Lam KS. Biochemistry. 1994;33:14825–14833.Meldal M, Svendsen I, Breddam K, Auzanneau F-I. Proc. Natl. Acad. Sci. U.S.A. 1994;91:3314–3318.Hu Y-J, Wei Y, Zhou Y, Rajagopalan PTR, Pei D. Biochemistry. 1999;38:643–650.Rosse G, Kueng E, Page MGP, Schauer-Vukasinovic V, Giller T, Lahm H-W, Hunziker P, Schlatter D. J. Comb. Chem. 2000;2:461–466.Garaud M, Pei D. J. Am. Chem. Soc. 2007;129:5366–5367..
-
-
-
For reviews see: Hoveyda AH. Chem. Biol. 1998;5:R187–R191.Revell JD, Wennerners H. Topics Curr. Chem. 2007;277:251–266..
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

