A new animal model of postsurgical bowel inflammation and fibrosis: the effect of commensal microflora
- PMID: 19398439
- PMCID: PMC2752281
- DOI: 10.1136/gut.2008.157636
A new animal model of postsurgical bowel inflammation and fibrosis: the effect of commensal microflora
Abstract
Objective: Ileocaecal resection (ICR) is common in Crohn's disease. Inflammation and fibrosis frequently recur at the site of anastomosis or in the small intestine (SI). No animal models of postsurgical inflammation and fibrosis exist. A model of ICR was developed in interleukin 10 (IL10) null and wild-type (WT) mice to test the hypothesis that ICR promotes postsurgical inflammation and fibrosis in the SI or anastomosis of genetically susceptible IL10 null, but not WT or germ-free (GF)-IL10 null mice.
Methods: GF-IL10 null mice were conventionalised (CONV) and 3 weeks later randomised to ICR, transection (T) or no treatment (NoTx). Age-matched conventionally raised (CONV) WT and GF-IL10 null mice received ICR, T or NoTx. Animals were killed 28 days later. Histological scoring, real-time PCR for tumour necrosis factor alpha and collagen, and immunostaining for CD3(+) T cells assessed inflammation and fibrosis.
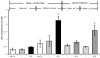
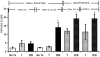
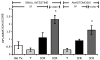
Results: After ICR, CONV-IL10 null, but not CONV-WT mice, developed significant inflammation and fibrosis in the SI and inflammation in anastomosis compared with NoTx or T controls. Fibrosis occurred in the anastomosis of both CONV-IL10 null and CONV-WT mice following ICR. GF-IL10 null mice developed little or no inflammation or fibrosis in the SI or anastomosis after ICR.
Conclusions: ICR in CONV-IL10 null mice provides a new animal model of postsurgical inflammation and fibrosis in the SI and anastomosis. Absence of inflammation and fibrosis in the SI of CONV-WT and GF-IL10 null mice following ICR indicates that postsurgical small bowel disease occurs only in genetically susceptible IL10 null mice and is bacteria dependent.
Figures



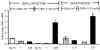

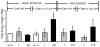

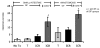

Similar articles
-
Inflammation enhances resection-induced intestinal adaptive growth in IL-10 null mice.J Surg Res. 2011 Jun 1;168(1):62-9. doi: 10.1016/j.jss.2009.09.051. Epub 2009 Oct 22. J Surg Res. 2011. PMID: 20074747 Free PMC article.
-
Small bowel fibrosis and systemic inflammatory response after ileocolonic anastomosis in IL-10 null mice.J Surg Res. 2012 Nov;178(1):147-54. doi: 10.1016/j.jss.2012.01.048. Epub 2012 Mar 31. J Surg Res. 2012. PMID: 22482772
-
Triptolide exerts protective effects against fibrosis following ileocolonic anastomosis by mechanisms involving the miR-16-1/HSP70 pathway in IL-10-deficient mice.Int J Mol Med. 2017 Aug;40(2):337-346. doi: 10.3892/ijmm.2017.3016. Epub 2017 Jun 9. Int J Mol Med. 2017. PMID: 28627592 Free PMC article.
-
Preventing Recurrence of Crohn's Disease Post-Ileocaecal Surgery in Paediatric Patients: A Therapy Guide Based on Systematic Review of the Evidence.Paediatr Drugs. 2024 Nov;26(6):659-672. doi: 10.1007/s40272-024-00650-w. Epub 2024 Aug 31. Paediatr Drugs. 2024. PMID: 39215954
-
[Surgery in Crohn's disease].Chirurg. 2015 Nov;86(11):1083-94. doi: 10.1007/s00104-015-0099-4. Chirurg. 2015. PMID: 26537846 Review. German.
Cited by
-
Localized intestinal radiation and liquid diet enhance survival and permit evaluation of long-term intestinal responses to high dose radiation in mice.PLoS One. 2012;7(12):e51310. doi: 10.1371/journal.pone.0051310. Epub 2012 Dec 7. PLoS One. 2012. PMID: 23236468 Free PMC article.
-
Impact of ileocecal resection and concomitant antibiotics on the microbiome of the murine jejunum and colon.PLoS One. 2013 Aug 27;8(8):e73140. doi: 10.1371/journal.pone.0073140. eCollection 2013. PLoS One. 2013. PMID: 24015295 Free PMC article.
-
Critical role of microbiota within cecal crypts on the regenerative capacity of the intestinal epithelium following surgical stress.Am J Physiol Gastrointest Liver Physiol. 2017 Feb 1;312(2):G112-G122. doi: 10.1152/ajpgi.00294.2016. Epub 2016 Dec 15. Am J Physiol Gastrointest Liver Physiol. 2017. PMID: 27979825 Free PMC article.
-
Inflammation enhances resection-induced intestinal adaptive growth in IL-10 null mice.J Surg Res. 2011 Jun 1;168(1):62-9. doi: 10.1016/j.jss.2009.09.051. Epub 2009 Oct 22. J Surg Res. 2011. PMID: 20074747 Free PMC article.
-
Yersiniabactin-Producing Adherent/Invasive Escherichia coli Promotes Inflammation-Associated Fibrosis in Gnotobiotic Il10-/- Mice.Infect Immun. 2019 Oct 18;87(11):e00587-19. doi: 10.1128/IAI.00587-19. Print 2019 Nov. Infect Immun. 2019. PMID: 31481410 Free PMC article.
References
-
- Loftus EV., Jr Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology. 2004;126:1504–17. - PubMed
-
- Hanauer SB. Inflammatory bowel disease: epidemiology, pathogenesis, and therapeutic opportunities. Inflamm Bowel Dis. 2006;12 (Suppl 1):S3–9. - PubMed
-
- Theiss AL, Fruchtman S, Lund PK. Growth factors in inflammatory bowel disease: the actions and interactions of growth hormone and insulin-like growth factor-I. Inflamm Bowel Dis. 2004;10:871–80. - PubMed
-
- Zimmermann EM, Lund PK. Fibrogenesis. Kirsner’s Inflammatory Bowel Disease. (6) 2004:219–29.
-
- Fichera A, Lovadina S, Rubin M, et al. Patterns and operative treatment of recurrent Crohn’s disease: a prospective longitudinal study. Surgery. 2006;140:649–54. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
