Sgt1 dimerization is negatively regulated by protein kinase CK2-mediated phosphorylation at Ser361
- PMID: 19398558
- PMCID: PMC2707205
- DOI: 10.1074/jbc.M109.012732
Sgt1 dimerization is negatively regulated by protein kinase CK2-mediated phosphorylation at Ser361
Abstract
The kinetochore, which consists of centromere DNA and structural proteins, is essential for proper chromosome segregation in eukaryotes. In budding yeast, Sgt1 and Hsp90 are required for the binding of Skp1 to Ctf13 (a component of the core kinetochore complex CBF3) and therefore for the assembly of CBF3. We have previously shown that Sgt1 dimerization is important for this kinetochore assembly mechanism. In this study, we report that protein kinase CK2 phosphorylates Ser(361) on Sgt1, and this phosphorylation inhibits Sgt1 dimerization.
Figures



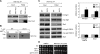
References
-
- Cheeseman I. M., Desai A. (2008) Nat. Rev. Mol. Cell Biol. 9, 33–46 - PubMed
-
- Westermann S., Drubin D. G., Barnes G. (2007) Annu. Rev. Biochem. 76, 563–571 - PubMed
-
- Sudakin V., Yen T. J. (2007) Biodrugs 21, 225–233 - PubMed
-
- Weaver B. A., Silk A. D., Montagna C., Verdier-Pinard P., Cleveland D. W. (2007) Cancer Cell 11, 25–36 - PubMed
-
- Fitzgerald-Hayes M., Clarke L., Carbon J. (1982) Cell 29, 235–244 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

