Appetite responds to changes in meal content, whereas ghrelin, leptin, and insulin track changes in energy availability
- PMID: 19401375
- PMCID: PMC2708957
- DOI: 10.1210/jc.2008-2495
Appetite responds to changes in meal content, whereas ghrelin, leptin, and insulin track changes in energy availability
Abstract
Context: It is uncertain how between-meal variations in energy availability and physiological changes in ghrelin, leptin, and insulin affect appetite.
Objective: The aim of the study was to examine the influence on human appetite of the meal size and its nutrient content or changes in energy availability and concentrations of ghrelin, leptin, and insulin.
Design: We conducted a crossover study manipulating meal size and energy availability through exercise energy expenditure and iv nutrient replacement (TPN).
Setting: The study was performed at a Clinical Research Center.
Participants: Ten healthy postmenopausal women (age, 59.7 +/- 1.5 yr; mean body mass index, 26 kg/m(2)) were studied.
Interventions: We conducted trials based on different morning meal size (418 vs. 2090 KJ), presence or absence of exercise energy expenditure (2273 to 2361 KJ), energy replacement by TPN (1521 to 1538 KJ), and a midday ad libitum meal.
Main outcome measures: Changes in hunger, fullness, midday ad libitum food consumption, and concentrations of ghrelin, leptin, insulin, and metabolic fuels were measured. We also performed midday meal tests for the presence of caloric compensation.
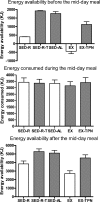
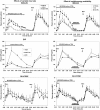
Results: Appetite was influenced by the size and energy content of the meals, but not by variation in energy availability which also did not trigger consummatory compensation. Exercise reduced hunger and increased fullness. Ghrelin, leptin, and insulin responded to changes in energy availability but not to meal size. Appetite was unaffected by physiological changes in ghrelin, leptin, or insulin.
Conclusions: During rest, appetite is influenced by the size and energy content of meals, but it bears no homeostatic relationship to between-meal changes in energy availability due to small meals, exercise, or TPN, or concentrations of ghrelin, leptin, and insulin.
Figures



Similar articles
-
Circadian and ultradian components of hunger in human non-homeostatic meal-to-meal eating.Physiol Behav. 2013 Oct 2;122:8-16. doi: 10.1016/j.physbeh.2013.08.001. Epub 2013 Aug 22. Physiol Behav. 2013. PMID: 23973366
-
Appetite, appetite hormone and energy intake responses to two consecutive days of aerobic exercise in healthy young men.Appetite. 2015 Sep;92:57-65. doi: 10.1016/j.appet.2015.05.006. Epub 2015 May 8. Appetite. 2015. PMID: 25963104 Clinical Trial.
-
The effect of meal frequency in a reduced-energy regimen on the gastrointestinal and appetite hormones in patients with type 2 diabetes: A randomised crossover study.PLoS One. 2017 Apr 3;12(4):e0174820. doi: 10.1371/journal.pone.0174820. eCollection 2017. PLoS One. 2017. PMID: 28369078 Free PMC article. Clinical Trial.
-
Is glycemic index of food a feasible predictor of appetite, hunger, and satiety?J Nutr Sci Vitaminol (Tokyo). 2009 Jun;55(3):201-7. doi: 10.3177/jnsv.55.201. J Nutr Sci Vitaminol (Tokyo). 2009. PMID: 19602827 Review.
-
Nonhomeostatic control of human appetite and physical activity in regulation of energy balance.Exerc Sport Sci Rev. 2010 Jul;38(3):114-21. doi: 10.1097/JES.0b013e3181e3728f. Exerc Sport Sci Rev. 2010. PMID: 20577059 Free PMC article. Review.
Cited by
-
Timing of Meals and Exercise Affects Hormonal Control of Glucoregulation, Insulin Resistance, Substrate Metabolism, and Gastrointestinal Hormones, but Has Little Effect on Appetite in Postmenopausal Women.Nutrients. 2021 Dec 1;13(12):4342. doi: 10.3390/nu13124342. Nutrients. 2021. PMID: 34959894 Free PMC article.
-
Nutritional Recommendations for Physique Athletes.J Hum Kinet. 2020 Jan 31;71:79-108. doi: 10.2478/hukin-2019-0096. eCollection 2020 Jan. J Hum Kinet. 2020. PMID: 32148575 Free PMC article.
-
After the spotlight: are evidence-based recommendations for refeeding post-contest energy restriction available for physique athletes? A scoping review.J Int Soc Sports Nutr. 2022 Aug 8;19(1):505-528. doi: 10.1080/15502783.2022.2108333. eCollection 2022. J Int Soc Sports Nutr. 2022. PMID: 35966021 Free PMC article.
-
Are gastrointestinal signals the principal guides to human appetite and energy balance?Med Res Arch. 2023 Jan;11(1):10.18103/mra.v11i1.3548. doi: 10.18103/mra.v11i1.3548. Epub 2023 Jan 31. Med Res Arch. 2023. PMID: 37234789 Free PMC article.
-
Serum leptin levels and reproductive function during the menstrual cycle.Am J Obstet Gynecol. 2014 Mar;210(3):248.e1-9. doi: 10.1016/j.ajog.2013.11.009. Epub 2013 Nov 8. Am J Obstet Gynecol. 2014. PMID: 24215851 Free PMC article.
References
-
- Batterham RL, Cowley MA, Small CJ, Herzog H, Cohen MA, Dakin CL, Wren AM, Brynes AE, Low MJ, Ghatei MA, Cone RD, Bloom SR 2002 Gut hormone PYY(3–36) physiologically inhibits food intake. Nature 418:650–654 - PubMed
-
- Woods SC 2004 Gastrointestinal satiety signals. An overview of gastrointestinal signals that influence food intake. Am J Physiol Gastrointest Liver Physiol 286:G7–G13 - PubMed
-
- Cummings DE, Purnell JQ, Frayo RS, Schmidova K, Wisse BE, Weigle DS 2001 A preprandial rise in plasma ghrelin levels suggests a role in meal initiation in humans. Diabetes 50:1714–1719 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

