Topoisomerase II alpha and responsiveness of breast cancer to adjuvant chemotherapy
- PMID: 19401546
- PMCID: PMC2677575
- DOI: 10.1093/jnci/djp067
Topoisomerase II alpha and responsiveness of breast cancer to adjuvant chemotherapy
Abstract
Background: Amplification or deletion of the topoisomerase II alpha (TOP2A) gene in breast cancers has been postulated to be more closely associated with responsiveness to anthracycline-containing chemotherapy than amplification of the human epidermal growth factor receptor type 2 (HER2) gene.
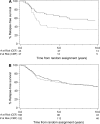
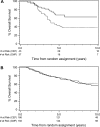
Methods: We studied 438 tumors from 710 premenopausal women with node-positive breast cancer who received cyclophosphamide, epirubicin, and 5-fluorouracil (CEF) or cyclophosphamide, methotrexate, and 5-fluorouracil (CMF) as adjuvant chemotherapy in the randomized National Cancer Institute of Canada Clinical Trials Group Mammary 5 (MA.5) trial. TOP2A alterations and HER2 amplification were quantified by fluorescence in situ hybridization. The association of TOP2A and HER2 status with recurrence-free survival (RFS) and overall survival (OS) in the two treatment groups was analyzed using Kaplan-Meier curves, the log-rank test, and Cox proportional hazard models. All statistical tests were two-sided.
Results: In patients whose tumors showed TOP2A alterations (either amplifications or deletions), treatment with CEF was statistically significantly superior to treatment with CMF in terms of RFS (adjusted hazard ratio [HR] = 0.35, 95% confidence interval [CI] = 0.17 to 0.73, P = .005) and OS (adjusted HR = 0.33, 95% CI = 0.15 to 0.75, P = .008). In patients without TOP2A amplification or deletion, the corresponding adjusted hazard ratios for RFS and OS were 0.90 (95% CI = 0.66 to 1.23, P = .49) and 1.09 (95% CI = 0.77 to 1.56, P = .62). Adjusted tests of interaction between treatment and TOP2A status were P = .09 for RFS and P = .02 for OS. Adjusted tests of interaction between treatment and HER2 status were P = .008 for RFS and P = .02 for OS.
Conclusion: TOP2A gene alterations (amplifications or deletions) are associated with an increase in responsiveness to anthracycline-containing chemotherapy regimens relative to non-anthracycline regimens that is similar to that seen in patients with HER2 amplification.
Figures
Comment in
-
Alterations in the TOP2A and HER2 genes: association with adjuvant anthracycline sensitivity in human breast cancers.J Natl Cancer Inst. 2009 May 6;101(9):615-8. doi: 10.1093/jnci/djp092. Epub 2009 Apr 28. J Natl Cancer Inst. 2009. PMID: 19401550 No abstract available.
-
Re: Topoisomerase II alpha and responsiveness of breast cancer to adjuvant chemotherapy.J Natl Cancer Inst. 2009 Dec 16;101(24):1735-6; author reply 1736-7. doi: 10.1093/jnci/djp402. J Natl Cancer Inst. 2009. PMID: 19893008 No abstract available.
Similar articles
-
Topoisomerase II alpha protein and responsiveness of breast cancer to adjuvant chemotherapy with CEF compared to CMF in the NCIC CTG randomized MA.5 adjuvant trial.Breast Cancer Res Treat. 2011 Jul;128(2):401-9. doi: 10.1007/s10549-011-1511-5. Epub 2011 Apr 26. Breast Cancer Res Treat. 2011. PMID: 21519837 Clinical Trial.
-
HER2 and TOP2A as predictive markers for anthracycline-containing chemotherapy regimens as adjuvant treatment of breast cancer: a meta-analysis of individual patient data.Lancet Oncol. 2011 Nov;12(12):1134-42. doi: 10.1016/S1470-2045(11)70231-5. Epub 2011 Sep 12. Lancet Oncol. 2011. PMID: 21917518
-
retrospective analysis of topoisomerase IIa amplifications and deletions as predictive markers in primary breast cancer patients randomly assigned to cyclophosphamide, methotrexate, and fluorouracil or cyclophosphamide, epirubicin, and fluorouracil: Danish Breast Cancer Cooperative Group.J Clin Oncol. 2005 Oct 20;23(30):7483-90. doi: 10.1200/JCO.2005.11.007. J Clin Oncol. 2005. PMID: 16234514 Clinical Trial.
-
Assessment of Topoisomerase II α status in breast cancer by quantitative PCR, gene expression microarrays, immunohistochemistry, and fluorescence in situ hybridization.Am J Pathol. 2011 Apr;178(4):1453-60. doi: 10.1016/j.ajpath.2010.12.042. Am J Pathol. 2011. PMID: 21435434 Free PMC article. Review.
-
Adjuvant chemotherapy in early breast cancer.Dan Med J. 2016 May;63(5):B5222. Dan Med J. 2016. PMID: 27127018 Review.
Cited by
-
Responsiveness of intrinsic subtypes to adjuvant anthracycline substitution in the NCIC.CTG MA.5 randomized trial.Clin Cancer Res. 2012 Apr 15;18(8):2402-12. doi: 10.1158/1078-0432.CCR-11-2956. Epub 2012 Feb 20. Clin Cancer Res. 2012. PMID: 22351696 Free PMC article. Clinical Trial.
-
Integrating genetics and epigenetics in breast cancer: biological insights, experimental, computational methods and therapeutic potential.BMC Syst Biol. 2015 Sep 21;9:62. doi: 10.1186/s12918-015-0211-x. BMC Syst Biol. 2015. PMID: 26391647 Free PMC article. Review.
-
Topoisomerase II α Gene alteration in Triple Negative Breast Cancer and Its Predictive Role for Anthracycline-Based Chemotherapy (Egyptian NCI Patients).Asian Pac J Cancer Prev. 2018 Dec 25;19(12):3581-3589. doi: 10.31557/APJCP.2018.19.12.3581. Asian Pac J Cancer Prev. 2018. PMID: 30583686 Free PMC article.
-
Role of Topoisomerases in Pediatric High Grade Osteosarcomas: TOP2A Gene Is One of the Unique Molecular Biomarkers of Chemoresponse.Cancers (Basel). 2013 Jun 4;5(2):662-75. doi: 10.3390/cancers5020662. Cancers (Basel). 2013. PMID: 24216996 Free PMC article.
-
HER2 and TOP2A in high-risk early breast cancer patients treated with adjuvant epirubicin-based dose-dense sequential chemotherapy.J Transl Med. 2012 Jan 12;10:10. doi: 10.1186/1479-5876-10-10. J Transl Med. 2012. PMID: 22240029 Free PMC article.
References
-
- Pritchard KI, Shepherd LE, O’Malley FP, et al. HER2 and responsiveness of breast cancer to adjuvant chemotherapy. N Engl J Med. 2006;354(20):2103–2111. - PubMed
-
- Dhesy-Thind B, Pritchard K, Messersmith H, O’Malley F, Elavathil L, Trudeau M. HER2/neu in systemic therapy for women with breast cancer: a systematic review. Breast Cancer Res Treat. 2008;109(5):209–229. - PubMed
-
- De Laurentiis M, Arpino G, Massarelli E, et al. A meta-analysis on the interaction between HER-2 expression and response to endocrine treatment in advanced breast cancer. Clin Cancer Res. 2005;11(13):4741–4748. - PubMed
-
- Gennari A, Sormani MP, Pronzato P, et al. HER2 status and efficacy of adjuvant anthracyclines in early breast cancer: a pooled analysis of randomized trials. J Natl Cancer Inst. 2008;100(1):14–20. - PubMed
-
- Tanner M, Isola J, Wiklund T, et al. Topoisomerase II alpha gene amplification predicts favorable treatment response to tailored and dose-escalated anthracycline-based adjuvant chemotherapy in HER-2/neu-amplified breast cancer: Scandinavian Breast Group Trial 9401. J Clin Oncol. 2006;24(16):2428–2436. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

