Jugular venous overflow of noradrenaline from the brain: a neurochemical indicator of cerebrovascular sympathetic nerve activity in humans
- PMID: 19403604
- PMCID: PMC2714023
- DOI: 10.1113/jphysiol.2008.167999
Jugular venous overflow of noradrenaline from the brain: a neurochemical indicator of cerebrovascular sympathetic nerve activity in humans
Abstract
A novel neurochemical method was applied for studying the activity of sympathetic nerves in the human cerebral vascular system. The aim was to investigate whether noradrenaline plasma kinetic measurements made with internal jugular venous sampling reflect cerebrovascular sympathetic activity. A database was assembled of fifty-six healthy subjects in whom total body noradrenaline spillover (indicative of whole body sympathetic nervous activity), brain noradrenaline spillover and brain lipophlic noradrenaline metabolite (3,4-dihydroxyphenolglycol (DHPG) and 3-methoxy-4-hydroxyphenylglycol (MHPG)) overflow rates were measured. These measurements were also made following ganglion blockade (trimethaphan, n = 6), central sympathetic inhibition (clonidine, n = 4) and neuronal noradrenaline uptake blockade (desipramine, n = 13) and in a group of patients (n = 9) with pure autonomic failure (PAF). The mean brain noradrenline spillover and brain noradrenaline metabolite overflow in healthy subjects were 12.5 +/- 1.8, and 186.4 +/- 25 ng min(-1), respectively, with unilateral jugular venous sampling for both. Total body noradrenaline spillover was 605.8 ng min(-1) +/- 34.4 ng min(-1). As expected, trimethaphan infusion lowered brain noradrenaline spillover (P = 0.03), but perhaps surprisingly increased jugular overflow of brain metabolites (P = 0.01). Suppression of sympathetic nervous outflow with clonidine lowered brain noradrenaline spillover (P = 0.004), without changing brain metabolite overflow (P = 0.3). Neuronal noradrenaline uptake block with desipramine lowered the transcranial plasma extraction of tritiated noradrenaline (P = 0.001). The PAF patients had 77% lower brain noradrenaline spillover than healthy recruits (P = 0.06), indicating that in them sympathetic nerve degeneration extended to the cerebral circulation, but metabolites overflow was similar to healthy subjects (P = 0.3). The invariable discordance between noradrenline spillover and noradrenaline metabolite overflow from the brain under these different circumstances indicates that the two measures arise from different sources, i.e. noradrenaline spillover originates from the cerebral vasculature outside the blood-brain barrier, and the noradrenaline metabolites originate primarily from brain noradrenergic neurons. We suggest that measurements of transcranial plasma noradrenaline spillover have utility as a method for assessing the sympathetic nerve activity of the cerebral vasculature.
Figures

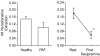
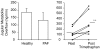
References
-
- Aggarwal A, Esler MD, Morris MJ, Lambert G, Kaye DM. Regional sympathetic effects of low-dose clonidine in heart failure. Hypertension. 2003;41:553–557. - PubMed
-
- Cox HS, Kaye DM, Thompson JM. Regional sympathetic nervous activation after a large meal in humans. Clin Sci. 1995;89:145–154. - PubMed
-
- Edvinnson L, MacKenzie ET, Mcculloch J. Cerebral Blood Flow and Metabolism. New York: Raven Press; 1993.
-
- Eisenhofer G, Goldstein DS, Ropchak TG, Nguyen HQ, Keiser HR, Kopin IJ. Source and physiological significance of plasma 3,4-dihydroxyphenolglycol and 3-methoxy-4- hydroxyphenylglycol. J Auton Nerv Syst. 1988;24:1–14. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

