New insights into {gamma}-aminobutyric acid catabolism: Evidence for {gamma}-hydroxybutyric acid and polyhydroxybutyrate synthesis in Saccharomyces cerevisiae
- PMID: 19411412
- PMCID: PMC2704823
- DOI: 10.1128/AEM.00051-09
New insights into {gamma}-aminobutyric acid catabolism: Evidence for {gamma}-hydroxybutyric acid and polyhydroxybutyrate synthesis in Saccharomyces cerevisiae
Abstract
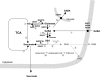
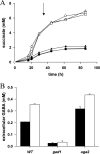
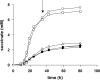
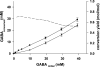
The gamma-aminobutyrate (GABA) shunt, an alternative route for the conversion of alpha-ketoglutarate to succinate, involves the glutamate decarboxylase Gad1p, the GABA transaminase Uga1p and the succinate semialdehyde dehydrogenase Uga2p. This pathway has been extensively described in plants and animals, but its function in yeast remains unclear. We show that the flux through Gad1p is insignificant during fermentation in rich sugar-containing medium, excluding a role for this pathway in redox homeostasis under anaerobic conditions or sugar stress. However, we found that up to 4 g of exogenous GABA/liter was efficiently consumed by yeast. We studied the fate of this consumed GABA. Most was converted into succinate, with a reaction yield of 0.7 mol/mol. We also showed that a large proportion of GABA was stored within cells, indicating a possible role for this molecule in stress tolerance mechanisms or nitrogen storage. Furthermore, based on enzymatic and metabolic evidence, we identified an alternative route for GABA catabolism, involving the reduction of succinate-semialdehyde into gamma-hydroxybutyric acid and the polymerization of gamma-hydroxybutyric acid to form poly-(3-hydroxybutyric acid-co-4-hydroxybutyric acid). This study provides the first demonstration of a native route for the formation of this polymer in yeast. Our findings shed new light on the GABA pathway and open up new opportunities for industrial applications.
Figures


References
-
- Allan, W. L., R. W. Smith, and B. J. Shelp. 2003. Direct measurement of gamma-hydroxybutyrate (GBH) in crude plant extracts by liquid chromatography/negative ion electrospray: mass spectrometry. Application bulletin no. AB-0015:4. Agilent Technologies, Inc., Mississauga, Ontario, Canada.
-
- Andre, B., C. Hein, M. Grenson, and J. C. Jauniaux. 1993. Cloning and expression of the UGA4 gene coding for the inducible GABA-specific transport protein of Saccharomyces cerevisiae. Mol. Gen. Genet. 237:17-25. - PubMed
-
- Bely, M., J. M. Sablayrolles, and P. Barre. 1990. Description of alcoholic fermentation kinetics: its variability and significance. Am. J. Enol. Vitic. 41:319-324.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

