Vernalization-induced flowering in cereals is associated with changes in histone methylation at the VERNALIZATION1 gene
- PMID: 19416817
- PMCID: PMC2677093
- DOI: 10.1073/pnas.0903566106
Vernalization-induced flowering in cereals is associated with changes in histone methylation at the VERNALIZATION1 gene
Abstract
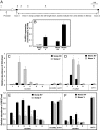
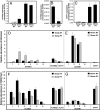
Prolonged exposure to low temperatures (vernalization) accelerates the transition to reproductive growth in many plant species, including the model plant Arabidopsis thaliana and the economically important cereal crops, wheat and barley. Vernalization-induced flowering is an epigenetic phenomenon. In Arabidopsis, stable down-regulation of FLOWERING LOCUS C (FLC) by vernalization is associated with changes in histone modifications at FLC chromatin. In cereals, the vernalization response is mediated by stable induction of the floral promoter VERNALIZATION1 (VRN1), which initiates reproductive development at the shoot apex. We show that in barley (Hordeum vulgare), repression of HvVRN1 before vernalization is associated with high levels of histone 3 lysine 27 trimethylation (H3K27me3) at HvVRN1 chromatin. Vernalization caused increased levels of histone 3 lysine 4 trimethylation (H3K4me3) and a loss of H3K27me3 at HvVRN1, suggesting that vernalization promotes an active chromatin state at VRN1. Levels of these histone modifications at 2 other flowering-time genes, VERNALIZATION2 and FLOWERING LOCUS T, were not altered by vernalization. Our study suggests that maintenance of an active chromatin state at VRN1 is likely to be the basis for epigenetic memory of vernalization in cereals. Thus, regulation of chromatin state is a feature of epigenetic memory of vernalization in Arabidopsis and the cereals; however, whereas vernalization-induced flowering in Arabidopsis is mediated by epigenetic regulation of the floral repressor FLC, this phenomenon in cereals is mediated by epigenetic regulation of the floral activator, VRN1.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Sung S, Amasino R. Remembering winter: Toward a molecular understanding of vernalization. Ann Rev Plant Biol. 2005;56:491–508. - PubMed
-
- Trevaskis B, Hemming MN, Dennis ES, Peacock WJ. The molecular basis of vernalization-induced flowering in cereals. Trends Plants Sci. 2007;12:352–357. - PubMed
-
- Helliwell CA, Wood CC, Robertson M, Peacock WJ, Dennis ES. The Arabidopsis FLC protein interacts directly in vivo with SOC1 and FT chromatin and is part of a high molecular-weight protein complex. Plant J. 2006;46:83–192. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

