The pannexin 1 channel activates the inflammasome in neurons and astrocytes
- PMID: 19416975
- PMCID: PMC2709345
- DOI: 10.1074/jbc.M109.004804
The pannexin 1 channel activates the inflammasome in neurons and astrocytes
Abstract
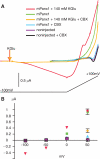
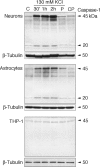
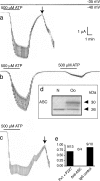
The inflammasome is a multiprotein complex involved in innate immunity. Activation of the inflammasome causes the processing and release of the cytokines interleukins 1beta and 18. In primary macrophages, potassium ion flux and the membrane channel pannexin 1 have been suggested to play roles in inflammasome activation. However, the molecular mechanism(s) governing inflammasome signaling remains poorly defined, and it is undetermined whether these mechanisms apply to the central nervous system. Here we show that high extracellular potassium opens pannexin channels leading to caspase-1 activation in primary neurons and astrocytes. The effect of K(+) on pannexin 1 channels was independent of membrane potential, suggesting that stimulation of inflammasome signaling was mediated by an allosteric effect. The activation of the inflammasome by K(+) was inhibited by the pannexin 1 channel blocker probenecid, supporting a role of pannexin 1 in inflammasome activation. Co-immunoprecipitation of neuronal lysates indicates that pannexin 1 associates with components of the multiprotein inflammasome complex, including the P2X7 receptor and caspase-1. Moreover antibody neutralization of the adaptor protein ASC (apoptosis-associated speck-like protein containing a CARD) blocked ATP-induced cell death in oocytes co-expressing P2X7 receptor and pannexin 1. Thus, in contrast to macrophages and monocytes in which low intracellular K(+) has been suggested to trigger inflammasome activation, in neural cells, high extracellular K(+) activates caspase-1 probably through pannexin 1.
Figures

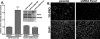



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

