Hypoxia limits antioxidant capacity in red blood cells by altering glycolytic pathway dominance
- PMID: 19417084
- PMCID: PMC2735367
- DOI: 10.1096/fj.09-130666
Hypoxia limits antioxidant capacity in red blood cells by altering glycolytic pathway dominance
Abstract
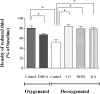
The erythrocyte membrane is a newly appreciated platform for thiol-based circulatory signaling, and it requires robust free thiol maintenance. We sought to define physiological constraints on erythrocyte antioxidant defense. Hemoglobin (Hb) conformation gates glycolytic flux through the hexose monophosphate pathway (HMP), the sole source of nicotinamide adenine dinucleotide phosphate (NADPH) in erythrocytes. We hypothesized elevated intraerythrocytic deoxyHb would limit resilience to oxidative stress. Human erythrocytes were subjected to controlled oxidant (superoxide) loading following independent manipulation of oxygen tension, Hb conformation, and glycolytic pathway dominance. Sufficiency of antioxidant defense was determined by serial quantification of GSH, NADPH, NADH redox couples. Hypoxic erythrocytes demonstrated greater loss of reduction potential [Delta GSH E(hc) (mV): 123.4+/-9.7 vs. 57.2+/-11.1] and reduced membrane thiol (47.7+/-5.7 vs. 20.1+/-4.3%) (hypoxia vs. normoxia, respectively; P<0.01), a finding mimicked in normoxic erythrocytes after HMP blockade. Rebalancing HMP flux during hypoxia restored resilience to oxidative stress at all stages of the system. Cell-free studies assured oxidative loading was not altered by oxygen tension, heme ligation, or the inhibitors employed. These data indicate that Hb conformation controls coupled glucose and thiol metabolism in erythrocytes, and implicate hypoxemia in the pathobiology of erythrocyte-based vascular signaling.
Figures



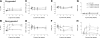
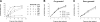


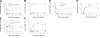
Similar articles
-
Downregulated Recycling Process but Not De Novo Synthesis of Glutathione Limits Antioxidant Capacity of Erythrocytes in Hypoxia.Oxid Med Cell Longev. 2020 Sep 4;2020:7834252. doi: 10.1155/2020/7834252. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 32963701 Free PMC article.
-
Quantifying dynamic range in red blood cell energetics: Evidence of progressive energy failure during storage.Transfusion. 2021 May;61(5):1586-1599. doi: 10.1111/trf.16395. Epub 2021 Apr 8. Transfusion. 2021. PMID: 33830505 Free PMC article.
-
Sickle hemoglobin disturbs normal coupling among erythrocyte O2 content, glycolysis, and antioxidant capacity.Blood. 2013 Feb 28;121(9):1651-62. doi: 10.1182/blood-2012-02-414037. Epub 2013 Jan 7. Blood. 2013. PMID: 23297128 Free PMC article.
-
Redox imbalance, macrocytosis, and RBC homeostasis.Antioxid Redox Signal. 2006 Jul-Aug;8(7-8):1205-16. doi: 10.1089/ars.2006.8.1205. Antioxid Redox Signal. 2006. PMID: 16910768 Review.
-
Proliferating tumor cells mimick glucose metabolism of mature human erythrocytes.Cell Cycle. 2019 Jun;18(12):1316-1334. doi: 10.1080/15384101.2019.1618125. Epub 2019 Jun 3. Cell Cycle. 2019. PMID: 31154896 Free PMC article. Review.
Cited by
-
The CYB5R3c .350C>G and G6PD A alleles modify severity of anemia in malaria and sickle cell disease.Am J Hematol. 2020 Nov;95(11):1269-1279. doi: 10.1002/ajh.25941. Epub 2020 Sep 23. Am J Hematol. 2020. PMID: 32697331 Free PMC article. Clinical Trial.
-
Storing red blood cells with vitamin C and N-acetylcysteine prevents oxidative stress-related lesions: a metabolomics overview.Blood Transfus. 2014 Jul;12(3):376-87. doi: 10.2450/2014.0266-13. Epub 2014 Jun 5. Blood Transfus. 2014. PMID: 25074788 Free PMC article. Clinical Trial.
-
Specific Etiologies Associated With the Multiple Organ Dysfunction Syndrome in Children: Part 2.Pediatr Crit Care Med. 2017 Mar;18(3_suppl Suppl 1):S58-S66. doi: 10.1097/PCC.0000000000001051. Pediatr Crit Care Med. 2017. PMID: 28248835 Free PMC article. Review.
-
Survival of red blood cells after transfusion: processes and consequences.Front Physiol. 2013 Dec 18;4:376. doi: 10.3389/fphys.2013.00376. Front Physiol. 2013. PMID: 24391593 Free PMC article. Review.
-
AltitudeOmics: Red Blood Cell Metabolic Adaptation to High Altitude Hypoxia.J Proteome Res. 2016 Oct 7;15(10):3883-3895. doi: 10.1021/acs.jproteome.6b00733. Epub 2016 Sep 27. J Proteome Res. 2016. PMID: 27646145 Free PMC article.
References
-
- Balagopalakrishna C, Manoharan P T, Abugo O O, Rifkind J M. Production of superoxide from hemoglobin-bound oxygen under hypoxic conditions. Biochemistry. 1996;35:6393–6398. - PubMed
-
- Droge W. Free radicals in the physiological control of cell function. Physiol Rev. 2002;82:47–95. - PubMed
-
- Gonzalez C, Sanz-Alfayate G, Agapito M T, Gomez-Nino A, Rocher A, Obeso A. Significance of ROS in oxygen sensing in cell systems with sensitivity to physiological hypoxia. Respir Physiol Neurobiol. 2002;132:17–41. - PubMed
-
- Buehler P W, Alayash A I. Oxygen sensing in the circulation: “cross talk” between red blood cells and the vasculature. Antioxidants Redox Signal. 2004;6:1000–1010. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

