Immune reconstitution inflammatory syndrome among HIV-infected South African infants initiating antiretroviral therapy
- PMID: 19417581
- PMCID: PMC2810152
- DOI: 10.1097/QAD.0b013e32832afefc
Immune reconstitution inflammatory syndrome among HIV-infected South African infants initiating antiretroviral therapy
Abstract
Objectives: To determine the incidence, clinical manifestations and risk factors for immune reconstitution inflammatory syndrome (IRIS) in young children initiating highly active antiretroviral therapy (HAART).
Design: A prospective cohort of antiretroviral-naïve HIV-infected children less than 24 months of age enrolled in a treatment strategies trial in Johannesburg, South Africa.
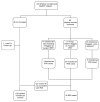
Methods: Among 169 HIV-infected children initiating HAART, April 2005 to November 2006, the records of 83 children suspected to have IRIS within 6 months of starting treatment were reviewed to determine whether they met criteria for IRIS. Seven were excluded due to incomplete follow-up. Pretreatment and post-treatment characteristics of children with and without IRIS were compared.
Results: Overall, 34/162 (21%) children developed IRIS at a median of 16 days (range 7-115 days) post-HAART initiation. Bacille Calmette-Guérin reaction was most common occurring in 24/34 (71%) children, primarily injection site lesions and/or ipsilateral axillary lymphadenitis with abscess. Other IRIS conditions (not mutually exclusive) included Mycobacterium tuberculosis (n = 12), cytomegalovirus pneumonia (n = 1), Streptococcus pneumonia sepsis (n = 1), and severe seborrheic dermatitis (n = 1). Children with IRIS were younger (median age 7 vs. 10 months, P = 0.007) with a lower CD4 cell percentage (median 13.9 vs. 19.2, P = 0.009) at HAART initiation than controls. After 24 weeks on HAART, 62% of IRIS cases vs. 28% of controls had HIV RNA more than 400 copies/ml (P = 0.001), odds ratio = 2.88 (95% confidence interval = 1.14-7.29) after adjusting for baseline factors.
Conclusion: Infants and young children with advanced HIV disease initiating HAART are at high risk for developing IRIS, leading to additional morbidity and possibly impairing virologic response to antiretroviral treatment.
Figures
References
-
- Murdoch DM, Venter DF, Feldman C, Van Rie A. Incidence and risk factors for the immune reconstitution inflammatory syndrome in HIV patients in South Africa: a prospective study. AIDS. 2008;22:601–610. - PubMed
-
- Kumarasamy N, Chaguturu S, Mayer KH, Solomon S, Yepthomi T, Balakrishnan P, Flanigan TP. Incidence of immune reconstitution syndrome in HIV/tuberculosis-coinfected patients after initiation of generic antiretroviral therapy in India. J Acquir Immune Defic Sydr. 2004;37:1574–1576. - PubMed
-
- Lawn SD, Bekker LG, Miller RF. Immune reconstitution disease associated with mycobacterial infections in HIV-infected individuals receiving antiretrovirals. Lancet Infect Dis. 2005;5:361–373. - PubMed
-
- Lawn SD, Myer L, Bekker LG, Wood R. Tuberculosis-associated immune reconstitution disease: incidence, risk factors and impact in an antiretroviral treatment service in South Africa. AIDS. 2007;21:335–341. - PubMed
-
- Shelburne SA, Montes M, Hammill RJ. Immune reconstitution inflammatory syndrome: more answers, more questions. J Antimicrob Chemother. 2006;57:167–170. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

