Serotonin excites fast-spiking interneurons in the striatum
- PMID: 19419423
- PMCID: PMC2695856
- DOI: 10.1111/j.1460-9568.2009.06725.x
Serotonin excites fast-spiking interneurons in the striatum
Abstract
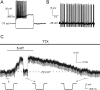
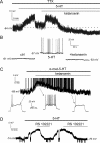
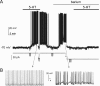
Fast-spiking interneurons (FSIs) control the output of the striatum by mediating feed-forward GABAergic inhibition of projection neurons. Their neuromodulation can therefore critically affect the operation of the basal ganglia. We studied the effects of 5-hydroxytryptamine (5-HT, serotonin), a neurotransmitter released in the striatum by fibres originating in the raphe nuclei, on FSIs recorded with whole-cell techniques in rat brain slices. Bath application of serotonin (30 microm) elicited slow, reversible depolarizations (9 +/- 3 mV) in 37/46 FSIs. Similar effects were observed using conventional whole-cell and gramicidin perforated-patch techniques. The serotonin effects persisted in the presence of tetrodotoxin and were mediated by 5-HT(2C) receptors, as they were reversed by the 5-HT(2) receptor antagonist ketanserin and by the selective 5-HT(2C) receptor antagonist RS 102221. Serotonin-induced depolarizations were not accompanied by a significant change in FSI input resistance. Serotonin caused the appearance of spontaneous firing in a minority (5/35) of responsive FSIs, whereas it strongly increased FSI excitability in each of the remaining responsive FSIs, significantly decreasing the latency of the first spike evoked by a current step and increasing spike frequency. Voltage-clamp experiments revealed that serotonin suppressed a current that reversed around -100 mV and displayed a marked inward rectification, a finding that explains the lack of effects of serotonin on input resistance. Consistently, the effects of serotonin were completely occluded by low concentrations of extracellular barium, which selectively blocks Kir2 channels. We concluded that the excitatory effects of serotonin on FSIs were mediated by 5-HT(2C) receptors and involved suppression of an inwardly rectifying K(+) current.
Figures


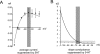
References
-
- Bevan MD, Magill PJ, Terman D, Bolam JP, Wilson CJ. Move to the rhythm: oscillations in the subthalamic nucleus-external globus pallidus network. Trends Neurosci. 2002;25:525–531. - PubMed
-
- Bonsi P, Cuomo D, Ding J, Sciamanna G, Ulrich S, Tscherter A, Bernardi G, Surmeier DJ, Pisani A. Endogenous serotonin excites striatal cholinergic interneurons via the activation of 5-HT 2C, 5-HT6, and 5-HT7 serotonin receptors: implications for extrapyramidal side effects of serotonin reuptake inhibitors. Neuropsychopharmacology. 2007;32:1840–1854. - PubMed

