Cholinergic optimization of cue-evoked parietal activity during challenged attentional performance
- PMID: 19419433
- PMCID: PMC3030246
- DOI: 10.1111/j.1460-9568.2009.06713.x
Cholinergic optimization of cue-evoked parietal activity during challenged attentional performance
Abstract
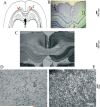
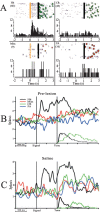
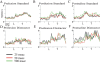
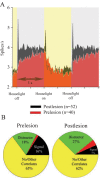
The detection of salient or instrumental stimuli and the selection of cue-evoked responses are mediated by a fronto-parietal network that is modulated by cholinergic inputs originating from the basal forebrain. Visual cues that guide behavior are more strongly represented in the posterior parietal cortex (PPC) than are similar cues that are missed or task-irrelevant. Although the crucial role of cholinergic inputs in cue detection has been demonstrated by lesion studies, the role of PPC neurons in the cholinergic modulation of cue detection is unclear. We recorded extracellular spikes from PPC neurons of rats performing a sustained attention task, before and after selective removal of cholinergic inputs to the recording site. Visual cues that were subsequently detected evoked significant increases in the PPC firing rate. In the absence of cholinergic input, the activation of PPC neurons by detected cues was greatly diminished. When a visual distractor was introduced during task performance, a population of PPC neurons selectively responded to the distractor. As a result of cholinergic deafferentation, distractor-related neuronal activity was enhanced, and the detection-related activity was further suppressed. Thus, in deafferented subjects, the distractor lowered the signal-to-noise ratio of cue-evoked responses. This impairment in cue-evoked neuronal activity may have mediated the increased response latencies observed for detected cues in the presence of the distractor. Additional experiments demonstrated that the effects of cholinergic deafferentation were not confounded by extended practice or electrode depth. Collectively, these findings indicate that cholinergic inputs to PPC neurons amplify cue detection, and may also act to suppress irrelevant distractors.
Figures




References
-
- Arnold HM, Burk JA, Sarter M, Bruno JP. Cortical acetylcholine release in rats performing an operant sustained attention task or operant control procedures. Neuroscience. 2002;114:451–460. - PubMed
-
- Bucci DJ, Macleod JE. Changes in neural activity associated with a surprising change in the predictive validity of a conditioned stimulus. Eur. J. Neurosci. 2007;26:2669–2676. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

