The monocyte chemoattractant protein-1/CCR2 loop, inducible by TGF-beta, increases podocyte motility and albumin permeability
- PMID: 19420107
- PMCID: PMC2711714
- DOI: 10.1152/ajprenal.90642.2008
The monocyte chemoattractant protein-1/CCR2 loop, inducible by TGF-beta, increases podocyte motility and albumin permeability
Abstract
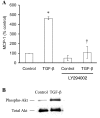
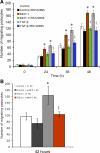
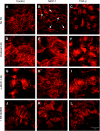
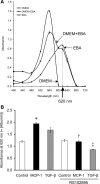
The role of monocyte chemoattractant protein-1 (MCP-1) in diabetic nephropathy is typically viewed through the lens of inflammation, but MCP-1 might exert noninflammatory effects on the kidney cells directly. Glomerular podocytes in culture, verified to express the marker nephrin, were exposed to diabetic mediators such as high glucose or angiotensin II and assayed for MCP-1. Only transforming growth factor-beta (TGF-beta) significantly increased MCP-1 production, which was prevented by SB431542 and LY294002, indicating that signaling proceeded through the TGF-beta type I receptor kinase and the phosphatidylinositol 3-kinase pathway. The TGF-beta-induced MCP-1 was found to activate the podocyte's cysteine-cysteine chemokine receptor 2 (CCR2) and, as a result, enhance the cellular motility, cause rearrangement of the actin cytoskeleton, and increase podocyte permeability to albumin in a Transwell assay. The preceding effects of TGF-beta were replicated by treatment with recombinant MCP-1 and blocked by a neutralizing anti-MCP-1 antibody or a specific CCR2 inhibitor, RS102895. In conclusion, this is the first description that TGF-beta signaling through PI3K induces the podocyte expression of MCP-1 that can then operate via CCR2 to increase cellular migration and alter albumin permeability characteristics. The pleiotropic effects of MCP-1 on the resident kidney cells such as the podocyte may exacerbate the disease process of diabetic albuminuria.
Figures




References
-
- Amann B, Tinzmann R, Angelkort B. ACE inhibitors improve diabetic nephropathy through suppression of renal MCP-1. Diabetes Care 26: 2421–2425, 2003. - PubMed
-
- Banba N, Nakamura T, Matsumura M, Kuroda H, Hattori Y, Kasai K. Possible relationship of monocyte chemoattractant protein-1 with diabetic nephropathy. Kidney Int 58: 684–690, 2000. - PubMed
-
- Berridge MV, Herst PM, Tan AS. Tetrazolium dyes as tools in cell biology: new insights into their cellular reduction. Biotechnol Annu Rev 11: 127–152, 2005. - PubMed
-
- Bondeva T, Ruster C, Franke S, Hammerschmid E, Klagsbrun M, Cohen CD, Wolf G. Advanced glycation end-products suppress neuropilin-1 expression in podocytes. Kidney Int 75: 605–616, 2009. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

