The anatomy and development of the claws of Xenopus laevis (Lissamphibia: Anura) reveal alternate pathways of structural evolution in the integument of tetrapods
- PMID: 19422431
- PMCID: PMC2736125
- DOI: 10.1111/j.1469-7580.2009.01052.x
The anatomy and development of the claws of Xenopus laevis (Lissamphibia: Anura) reveal alternate pathways of structural evolution in the integument of tetrapods
Abstract
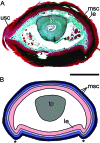
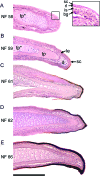
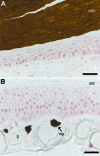
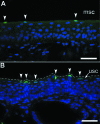
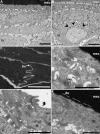
Digital end organs composed of hard, modified epidermis, generally referred to as claws, are present in mammals and reptiles as well as in several non-amniote taxa such as clawed salamanders and frogs, including Xenopus laevis. So far, only the claws and nails of mammals have been characterized extensively and the question of whether claws were present in the common ancestor of all extant tetrapods is as yet unresolved. To provide a basis for comparisons between amniote and non-amniote claws, we investigated the development, growth and ultrastructure of the epidermal component of the claws of X. laevis. Histological examination of developing claws of X. laevis shows that claw formation is initiated at the tip of the toe by the appearance of superficial cornified cells that are dark brown. Subsequent accumulation of new, proximally extended claw sheath corneocyte layers increases the length of the claw. Histological studies of adult claws show that proliferation of cornifying claw sheath cells occurs along the entire length of the claw-forming epidermis. Living epidermal cells that are converting into the cornified claw sheath corneocytes undergo a form of programmed cell death that is accompanied by degradation of nuclear DNA. Subsequently, the cytoplasm and the nuclear remnants acquire a brown colour by an as-yet unknown mechanism that is likely homologous to the colouration mechanism that occurs in other hard, cornified structures of amphibians such as nuptial pads and tadpole beaks. Transmission electron microscopy revealed that the cornified claw sheath consists of parallel layers of corneocytes with interdigitations being confined to intra-layer contacts and a cementing substance filling the intercorneocyte spaces. Together with recent reports that showed the main molecular components of amniote claws are absent in Xenopus, our data support the hypothesis that claws of amphibians likely represent clade-specific innovations, non-homologous to amniote claws.
Figures


Comment in
-
'The integument story: origins, evolution and current knowledge'.J Anat. 2009 Apr;214(4):407-8. doi: 10.1111/j.1469-7580.2009.01051.x. J Anat. 2009. PMID: 19422422 Free PMC article. No abstract available.
References
-
- Alibardi L. Keratinization in the epidermis of amphibians and the lungfish: comparison with amniote keratinization. Tissue Cell. 2001;33:439–449. - PubMed
-
- Alibardi L. Ultrastructural localization of histidine-labelled interkeratin matrix during keratinization of amphibian epidermis. Acta Histochemica. 2003;105:273–283. - PubMed
-
- Alibardi L. Microscopic analysis of lizard claw morphogenesis and hypothesis on its evolution. Acta Zool (Stockholm) 2008;89:169–178.
-
- Alibardi L, Toni M. Immuno-cross reactivity of Transglutaminase and cornification marker proteins in the epidermis of vertebrates suggests common processes of soft cornification across species. J Exp Zool (Mol Dev Evol) 2004;302B:526–549. - PubMed
-
- Alibardi L, Toni M, Valle LD. Hard cornification in reptilian epidermis in comparison to cornification in mammalian epidermis. Exp Dermatol. 2007;16:961–976. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

