3'-Phosphoadenosine-5'-phosphate phosphatase activity is required for superoxide stress tolerance in Streptococcus mutans
- PMID: 19429620
- PMCID: PMC2698468
- DOI: 10.1128/JB.00184-09
3'-Phosphoadenosine-5'-phosphate phosphatase activity is required for superoxide stress tolerance in Streptococcus mutans
Abstract
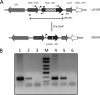
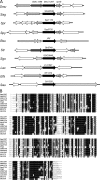
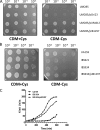
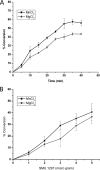
Aerobic microorganisms have evolved different strategies to withstand environmental oxidative stresses generated by various reactive oxygen species (ROS). For the facultative anaerobic human oral pathogen Streptococcus mutans, the mechanisms used to protect against ROS are not fully understood, since it does not possess catalase, an enzyme that degrades hydrogen peroxide. In order to elucidate the genes that are essential for superoxide stress response, methyl viologen (MV)-sensitive mutants of S. mutans were generated via ISS1 mutagenesis. Screening of approximately 2,500 mutants revealed six MV-sensitive mutants, each containing an insertion in one of five genes, including a highly conserved hypothetical gene, SMU.1297. Sequence analysis suggests that SMU.1297 encodes a hypothetical protein with a high degree of homology to the Bacillus subtilis YtqI protein, which possesses an oligoribonuclease activity that cleaves nano-RNAs and a phosphatase activity that degrades 3'-phosphoadenosine-5'-phosphate (pAp) and 3'-phosphoadenosine-5'-phosphosulfate (pApS) to produce AMP; the latter activity is similar to the activity of the Escherichia coli CysQ protein, which is required for sulfur assimilation. SMU.1297 was deleted using a markerless Cre-loxP-based strategy; the SMU.1297 deletion mutant was just as sensitive to MV as the ISS1 insertion mutant. Complementation of the deletion mutant with wild-type SMU.1297, in trans, restored the parental phenotype. Biochemical analyses with purified SMU.1297 protein demonstrated that it has pAp phosphatase activity similar to that of YtqI but apparently lacks an oligoribonuclease activity. The ability of SMU.1297 to dephosphorylate pApS in vivo was confirmed by complementation of an E. coli cysQ mutant with SMU.1297 in trans. Thus, our results suggest that SMU.1297 is involved in superoxide stress tolerance in S. mutans. Furthermore, the distribution of homologs of SMU.1297 in streptococci indicates that this protein is essential for superoxide stress tolerance in these organisms.
Figures



References
-
- Ajdic, D., W. M. McShan, R. E. McLaughlin, G. Savic, J. Chang, M. B. Carson, C. Primeaux, R. Tian, S. Kenton, H. Jia, S. Lin, Y. Qian, S. Li, H. Zhu, F. Najar, H. Lai, J. White, B. A. Roe, and J. J. Ferretti. 2002. Genome sequence of Streptococcus mutans UA159, a cariogenic dental pathogen. Proc. Natl. Acad. Sci. USA 9914434-14439. - PMC - PubMed
-
- Aravind, L., and E. V. Koonin. 1998. A novel family of predicted phosphoesterases includes Drosophila prune protein and bacterial RecJ exonuclease. Trends Biochem. Sci. 2317-19. - PubMed
-
- Banas, J. A. 2004. Virulence properties of Streptococcus mutans. Front. Biosci. 91267-1277. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

