Co-culture with mesenchymal stromal cells increases proliferation and maintenance of haematopoietic progenitor cells
- PMID: 19432817
- PMCID: PMC3837622
- DOI: 10.1111/j.1582-4934.2009.00776.x
Co-culture with mesenchymal stromal cells increases proliferation and maintenance of haematopoietic progenitor cells
Abstract
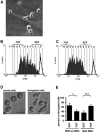
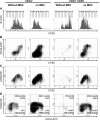
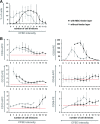
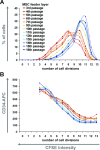
Mesenchymal stromal cells (MSC) have been suggested to provide a suitable cellular environment for in vitro expansion of haematopoietic stem and progenitor cells (HPC) from umbilical cord blood. In this study, we have simultaneously analysed the cell division history and immunophenotypic differentiation of HPC by using cell division tracking with carboxyfluorescein diacetate N-succinimidyl ester (CFSE). Co-culture with MSC greatly enhanced proliferation of human HPC, especially of the more primitive CD34(+)CD38(-) fraction. Without co-culture CD34 and CD133 expressions decreased after several cell divisions, whereas CD38 expression was up-regulated after some cell divisions and then diminished in fast proliferating cells. Co-culture with MSC maintained a primitive immunophenotype (CD34(+), CD133(+) and CD38(-)) for more population doublings, whereas up-regulation of differentiation markers (CD13, CD45 and CD56) in HPC was delayed to higher numbers of cell divisions. Especially MSC of early cell passages maintained CD34 expression in HPC over more cell divisions, whereas MSC of higher passages further enhanced their proliferation rate. Inhibition of mitogen-activated protein kinase 1 (MAPK1) impaired proliferation and differentiation of HPC, but not maintenance of long-term culture initiating cells. siRNA knockdown of N-cadherin and VCAM1 in feeder layer cells increased the fraction of slow dividing HPC, whereas knockdown of integrin beta 1 (ITGB1) and CD44 impaired their differentiation. In conclusion, MSC support proliferation as well as self-renewal of HPC with primitive immunophenotype. The use of early passages of MSC and genetic manipulation of proteins involved in HPC-MSC interaction might further enhance cord blood expansion on MSC.
Figures

References
-
- Da Silva CL, Goncalves R, Crapnell KB, et al. A human stromal-based serum-free culture system supports the ex vivo expansion/maintenance of bone marrow and cord blood hematopoietic stem/progenitor cells. Exp Hematol. 2005;33:828–35. - PubMed
-
- Hofmeister CC, Zhang J, Knight KL, et al. Ex vivo expansion of umbilical cord blood stem cells for transplantation: growing knowledge from the hematopoietic niche. Bone Marrow Transplant. 2007;39:11–23. - PubMed
-
- Kohler T, Plettig R, Wetzstein W, et al. Defining optimum conditions for the ex vivo expansion of human umbilical cord blood cells. Influences of progenitor enrichment, interference with feeder layers, early-acting cytokines and agitation of culture vessels. Stem Cells. 1999;17:19–24. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

