Up-regulation of uPARAP/Endo180 during culture activation of rat hepatic stellate cells and its presence in hepatic stellate cell lines from different species
- PMID: 19432973
- PMCID: PMC2689179
- DOI: 10.1186/1471-2121-10-39
Up-regulation of uPARAP/Endo180 during culture activation of rat hepatic stellate cells and its presence in hepatic stellate cell lines from different species
Abstract
Background: The urokinase plasminogen activator receptor associated protein (uPARAP)/Endo180 is a novel endocytic receptor that mediates collagen uptake and is implicated to play a role in physiological and pathological tissue-remodelling processes by mediating intracellular collagen degradation.
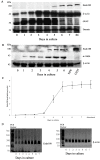
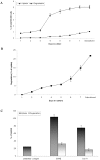
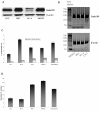
Result: This study investigates the expression of uPARAP/Endo180 protein and messenger RNA in primary rat hepatic stellate cell (HSC) cultures. The results show that uPARAP/Endo180 protein is not expressed in freshly isolated HSCs or during the first few days of culture while the cells still display quiescent features. In contrast, uPARAP/Endo180 protein is expressed early during HSC activation when cells are transdifferentiated into myofibroblast-like cells. Very low levels of uPARAP/Endo180 mRNA are detectable during the first days of culture but uPARAP/Endo180 mRNA is strongly up-regulated with increasing time in culture. Moreover, endocytic uptake of denatured collagen increases as transdifferentiation proceeds over time and correlates with increased expression of uPARAP/Endo180. Finally, analysis of uPARAP/Endo180 expression in four hepatic stellate cell lines from three different species showed that all these cell lines express uPARAP/Endo180 and are able to take up denatured collagen efficiently.
Conclusion: These results demonstrate that uPARAP/Endo180 expression by rat HSCs is strongly up-regulated during culture activation and identify this receptor as a feature common to culture-activated HSCs.
Figures

Similar articles
-
Uptake of denatured collagen into hepatic stellate cells: evidence for the involvement of urokinase plasminogen activator receptor-associated protein/Endo180.Biochem J. 2005 Apr 1;387(Pt 1):39-46. doi: 10.1042/BJ20040966. Biochem J. 2005. PMID: 15506989 Free PMC article.
-
Endocytic collagen degradation: a novel mechanism involved in protection against liver fibrosis.J Pathol. 2012 May;227(1):94-105. doi: 10.1002/path.3981. Epub 2012 Feb 17. J Pathol. 2012. PMID: 22294280
-
The collagen receptor uPARAP/Endo180 regulates collectins through unique structural elements in its FNII domain.J Biol Chem. 2020 Jul 3;295(27):9157-9170. doi: 10.1074/jbc.RA120.013710. Epub 2020 May 18. J Biol Chem. 2020. PMID: 32424040 Free PMC article.
-
The collagen receptor uPARAP/Endo180.Front Biosci (Landmark Ed). 2009 Jan 1;14(6):2103-14. doi: 10.2741/3365. Front Biosci (Landmark Ed). 2009. PMID: 19273187 Review.
-
uPARAP/Endo180: a multifaceted protein of mesenchymal cells.Cell Mol Life Sci. 2022 Apr 22;79(5):255. doi: 10.1007/s00018-022-04249-7. Cell Mol Life Sci. 2022. PMID: 35460056 Free PMC article. Review.
Cited by
-
Interaction of Lactobacillus fermentum BGHI14 with rat colonic mucosa: implications for colitis induction.Appl Environ Microbiol. 2013 Sep;79(18):5735-44. doi: 10.1128/AEM.01807-13. Epub 2013 Jul 12. Appl Environ Microbiol. 2013. PMID: 23851097 Free PMC article.
-
Endocytosis of collagen by hepatic stellate cells regulates extracellular matrix dynamics.Am J Physiol Cell Physiol. 2014 Oct 1;307(7):C622-33. doi: 10.1152/ajpcell.00086.2014. Epub 2014 Jul 30. Am J Physiol Cell Physiol. 2014. PMID: 25080486 Free PMC article.
-
GABA potentiate the immunoregulatory effects of Lactobacillus brevis BGZLS10-17 via ATG5-dependent autophagy in vitro.Sci Rep. 2020 Jan 28;10(1):1347. doi: 10.1038/s41598-020-58177-2. Sci Rep. 2020. PMID: 31992761 Free PMC article.
-
The collagen receptor uPARAP/Endo180 in tissue degradation and cancer (Review).Int J Oncol. 2015 Oct;47(4):1177-88. doi: 10.3892/ijo.2015.3120. Epub 2015 Aug 12. Int J Oncol. 2015. PMID: 26316068 Free PMC article. Review.
-
A novel functional role of collagen glycosylation: interaction with the endocytic collagen receptor uparap/ENDO180.J Biol Chem. 2011 Sep 16;286(37):32736-48. doi: 10.1074/jbc.M111.266692. Epub 2011 Jul 18. J Biol Chem. 2011. PMID: 21768090 Free PMC article.
References
-
- East L, Isacke CM. The mannose receptor family. Biochimica et biophysica acta. 2002;1572:364–386. - PubMed
-
- Sheikh H, Yarwood H, Ashworth A, Isacke CM. Endo180, an endocytic recycling glycoprotein related to the macrophage mannose receptor is expressed on fibroblasts, endothelial cells and macrophages and functions as a lectin receptor. Journal of cell science. 2000;113:1021–1032. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

