Isolation of stem/progenitor cells from normal lung tissue of adult humans
- PMID: 19438897
- PMCID: PMC6495959
- DOI: 10.1111/j.1365-2184.2009.00594.x
Isolation of stem/progenitor cells from normal lung tissue of adult humans
Abstract
Objectives: This study aimed to isolate and characterize stem/progenitor cells, starting from normal airway epithelia, obtained from human adults.
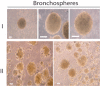
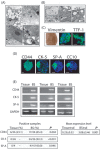
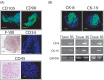
Materials and methods: Cultures of multicellular spheroids were obtained from human lung tissue specimens after mechanical and enzymatic digestion. Tissue-specific markers were detected on their cells by immunohistochemical and immunofluorescent techniques. Ultrastructural morphology of the spheroids (termed as bronchospheres) was evaluated by electron microscopy, gene expression analysis was performed by reverse transcription-polymerase chain reaction, and gene down-regulation was analysed by an RNA interference technique.
Results: Bronchospheres were found to be composed of cells with high expression of stem cell regulatory genes, which was not or was only weakly detectable in original tissues. Morphological analysis showed that bronchospheres were composed of mixed phenotype cells with type II alveolar and Clara cell features, highlighting their airway resident cell origin. In addition to displaying specific pulmonary and epithelial commitment, bronchospheres showed mesenchymal features. Silencing of the Slug gene, known to play a pivotal role in epithelial-mesenchymal transition processes and which was highly expressed in bronchospheres but not in original tissue, led bronchospheres to gain a differentiated bronchial/alveolar phenotype and to lose the stemness gene expression pattern.
Conclusions: Ours is the first study to describe ex vivo expansion of stem/progenitor cells resident in human lung epithelia, and our results suggest that the epithelial-mesenchymal transition process, still active in a subset of airway cells, may regulate transit of stem/progenitor cells towards epithelial differentiation.
Figures



Similar articles
-
Human Lung Spheroids as In Vitro Niches of Lung Progenitor Cells with Distinctive Paracrine and Plasticity Properties.Stem Cells Transl Med. 2017 Mar;6(3):767-777. doi: 10.5966/sctm.2015-0374. Epub 2016 Sep 22. Stem Cells Transl Med. 2017. PMID: 28297570 Free PMC article.
-
Isolation and Enrichment of Human Lung Epithelial Progenitor Cells for Organoid Culture.J Vis Exp. 2020 Jul 21;(161). doi: 10.3791/61541. J Vis Exp. 2020. PMID: 32773760
-
An Adult Mouse Thyroid Side Population Cell Line that Exhibits Enriched Epithelial-Mesenchymal Transition.Thyroid. 2017 Mar;27(3):460-474. doi: 10.1089/thy.2016.0130. Epub 2017 Feb 17. Thyroid. 2017. PMID: 28125936 Free PMC article.
-
[Advances in Classification and Research Methods of Lung Epithelial Stem and Progenitor Cells].Zhongguo Fei Ai Za Zhi. 2017 Feb 20;20(2):130-137. doi: 10.3779/j.issn.1009-3419.2017.02.08. Zhongguo Fei Ai Za Zhi. 2017. PMID: 28228225 Free PMC article. Review. Chinese.
-
Identification of novel resident pulmonary stem cells: form and function of the lung side population.Stem Cells. 2005 Sep;23(8):1073-81. doi: 10.1634/stemcells.2005-0039. Epub 2005 Jun 27. Stem Cells. 2005. PMID: 15987674 Review.
Cited by
-
Human iPS cell-derived alveolar epithelium repopulates lung extracellular matrix.J Clin Invest. 2013 Nov;123(11):4950-62. doi: 10.1172/JCI68793. Epub 2013 Oct 25. J Clin Invest. 2013. PMID: 24135142 Free PMC article.
-
Endogenous and exogenous stem cells: a role in lung repair and use in airway tissue engineering and transplantation.J Biomed Sci. 2010 Dec 7;17(1):92. doi: 10.1186/1423-0127-17-92. J Biomed Sci. 2010. PMID: 21138559 Free PMC article. Review.
-
Human Lung Spheroids as In Vitro Niches of Lung Progenitor Cells with Distinctive Paracrine and Plasticity Properties.Stem Cells Transl Med. 2017 Mar;6(3):767-777. doi: 10.5966/sctm.2015-0374. Epub 2016 Sep 22. Stem Cells Transl Med. 2017. PMID: 28297570 Free PMC article.
-
Proliferation Genes Repressed by TGF-β Are Downstream of Slug/Snail2 in Normal Bronchial Epithelial Progenitors and Are Deregulated in COPD.Stem Cell Rev Rep. 2021 Jun;17(3):703-718. doi: 10.1007/s12015-021-10123-z. Epub 2021 Jan 26. Stem Cell Rev Rep. 2021. PMID: 33495975
-
Lung organoids: current strategies for generation and transplantation.Cell Tissue Res. 2022 Dec;390(3):317-333. doi: 10.1007/s00441-022-03686-x. Epub 2022 Sep 30. Cell Tissue Res. 2022. PMID: 36178558 Free PMC article. Review.
References
-
- Jeffery PK, Hislop AA (2003) Embriology and growth In: Gibson GJ, Geddes DM, Costabel U, eds. Respir. Med., pp. 51–63. London: W.B. Saunders.
-
- Rawlins EL, Hogan BL (2006) Epithelial stem cells of the lung: privileged few or opportunities for many? Development 133, 2455–2465. - PubMed
-
- Borthwick DW, Shahbazian M, Krantz QT, Dorin JR, Randell SH (2001) Evidence for stem‐cell niches in the tracheal epithelium. Am. J. Respir. Cell Mol. Biol. 24, 662–670. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

