Voltage coupling of primary H+ V-ATPases to secondary Na+- or K+-dependent transporters
- PMID: 19448072
- PMCID: PMC2683009
- DOI: 10.1242/jeb.031534
Voltage coupling of primary H+ V-ATPases to secondary Na+- or K+-dependent transporters
Abstract
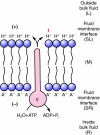
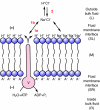
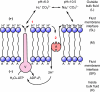
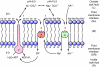
This review provides alternatives to two well established theories regarding membrane energization by H(+) V-ATPases. Firstly, we offer an alternative to the notion that the H(+) V-ATPase establishes a protonmotive force (pmf) across the membrane into which it is inserted. The term pmf, which was introduced by Peter Mitchell in 1961 in his chemiosmotic hypothesis for the synthesis of ATP by H(+) F-ATP synthases, has two parts, the electrical potential difference across the phosphorylating membrane, Deltapsi, and the pH difference between the bulk solutions on either side of the membrane, DeltapH. The DeltapH term implies three phases - a bulk fluid phase on the H(+) input side, the membrane phase and a bulk fluid phase on the H(+) output side. The Mitchell theory was applied to H(+) V-ATPases largely by analogy with H(+) F-ATP synthases operating in reverse as H(+) F-ATPases. We suggest an alternative, voltage coupling model. Our model for V-ATPases is based on Douglas B. Kell's 1979 'electrodic view' of ATP synthases in which two phases are added to the Mitchell model - an unstirred layer on the input side and another one on the output side of the membrane. In addition, we replace the notion that H(+) V-ATPases normally acidify the output bulk solution with the hypothesis, which we introduced in 1992, that the primary action of a H(+) V-ATPase is to charge the membrane capacitance and impose a Deltapsi across the membrane; the translocated hydrogen ions (H(+)s) are retained at the outer fluid-membrane interface by electrostatic attraction to the anions that were left behind. All subsequent events, including establishing pH differences in the outside bulk solution, are secondary. Using the surface of an electrode as a model, Kell's 'electrodic view' has five phases - the outer bulk fluid phase, an outer fluid-membrane interface, the membrane phase, an inner fluid-membrane interface and the inner bulk fluid phase. Light flash, H(+) releasing and binding experiments and other evidence provide convincing support for Kell's electrodic view yet Mitchell's chemiosmotic theory is the one that is accepted by most bioenergetics experts today. First we discuss the interaction between H(+) V-ATPase and the K(+)/2H(+) antiporter that forms the caterpillar K(+) pump, and use the Kell electrodic view to explain how the H(+)s at the outer fluid-membrane interface can drive two H(+) from lumen to cell and one K(+) from cell to lumen via the antiporter even though the pH in the bulk fluid of the lumen is highly alkaline. Exchange of outer bulk fluid K(+) (or Na(+)) with outer interface H(+) in conjunction with (K(+) or Na(+))/2H(+) antiport, transforms the hydrogen ion electrochemical potential difference, mu(H), to a K(+) electrochemical potential difference, mu(K) or a Na(+) electrochemical potential difference, mu(Na). The mu(K) or mu(Na) drives K(+)- or Na(+)-coupled nutrient amino acid transporters (NATs), such as KAAT1 (K(+) amino acid transporter 1), which moves Na(+) and an amino acid into the cell with no H(+)s involved. Examples in which the voltage coupling model is used to interpret ion and amino acid transport in caterpillar and larval mosquito midgut are discussed.
Figures
Similar articles
-
Cationic pathway of pH regulation in larvae of Anopheles gambiae.J Exp Biol. 2008 Mar;211(Pt 6):957-68. doi: 10.1242/jeb.012021. J Exp Biol. 2008. PMID: 18310121
-
Animal plasma membrane energization by chemiosmotic H+ V-ATPases.J Exp Biol. 1997 Jan;200(Pt 2):203-16. doi: 10.1242/jeb.200.2.203. J Exp Biol. 1997. PMID: 9050228 Review.
-
A vacuolar-type proton pump energizes K+/H+ antiport in an animal plasma membrane.J Biol Chem. 1991 Aug 15;266(23):15340-7. J Biol Chem. 1991. PMID: 1831202
-
NHE(VNAT): an H+ V-ATPase electrically coupled to a Na+:nutrient amino acid transporter (NAT) forms an Na+/H+ exchanger (NHE).J Exp Biol. 2009 Feb;212(Pt 3):347-57. doi: 10.1242/jeb.026047. J Exp Biol. 2009. PMID: 19151209 Free PMC article. Review.
-
Bacterial Na+ - or H+ -coupled ATP synthases operating at low electrochemical potential.Adv Microb Physiol. 2004;49:175-218. doi: 10.1016/S0065-2911(04)49004-3. Adv Microb Physiol. 2004. PMID: 15518831 Review.
Cited by
-
A Bacillus flagellar motor that can use both Na+ and K+ as a coupling ion is converted by a single mutation to use only Na+.PLoS One. 2012;7(9):e46248. doi: 10.1371/journal.pone.0046248. Epub 2012 Sep 25. PLoS One. 2012. PMID: 23049994 Free PMC article.
-
Transcellular and paracellular pathways of transepithelial fluid secretion in Malpighian (renal) tubules of the yellow fever mosquito Aedes aegypti.Acta Physiol (Oxf). 2011 Jul;202(3):387-407. doi: 10.1111/j.1748-1716.2010.02195.x. Epub 2010 Nov 16. Acta Physiol (Oxf). 2011. PMID: 20946239 Free PMC article. Review.
-
Kissing or fused since some time.Front Synaptic Neurosci. 2010 Sep 15;2:142. doi: 10.3389/fnsyn.2010.00142. eCollection 2010. Front Synaptic Neurosci. 2010. PMID: 21423528 Free PMC article. No abstract available.
-
Effects of Periplocoside P from Periploca sepium on the Midgut Transmembrane Potential of Mythimna separata Larvae.Sci Rep. 2016 Nov 11;6:36982. doi: 10.1038/srep36982. Sci Rep. 2016. PMID: 27833169 Free PMC article.
-
Electrical hypothesis of toxicity of the Cry toxins for mosquito larvae.Biosci Rep. 2013 Jan 11;33(1):125-36. doi: 10.1042/BSR20120101. Biosci Rep. 2013. PMID: 23083299 Free PMC article.
References
-
- Adams, M. D., Celniker, S. E., Holt, R. A., Evans, C. A., Gocayne, J. D., Amanatides, P. G., Scherer, S. E., Li, P. W., Hoskins, R. A., Galle, R. F. et al. (2000). The genome sequence of Drosophila melanogaster. Science 287, 2185-2195. - PubMed
-
- Azuma, M., Harvey, W. R. and Wieczorek, H. (1995). Stoichiometry of K+/H+ antiport helps to explain extracellular pH 11 in a model epithelium. FEBS Lett. 361, 153-156. - PubMed
-
- Beyenbach, K. W. (1995). Mechanisms and regulation of electrolyte transport in Malpighian tubules. J. Insect Physiol. 41, 197-207.
-
- Beyenbach, K. W. and Wieczorek, H. (2006). The V-type H+ ATPase: molecular structure and function, physiological roles and regulation. J. Exp. Biol. 209, 577-589. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

