Pharmacokinetics-pharmacodynamics of pyrazinamide in a novel in vitro model of tuberculosis for sterilizing effect: a paradigm for faster assessment of new antituberculosis drugs
- PMID: 19451303
- PMCID: PMC2715614
- DOI: 10.1128/AAC.01681-08
Pharmacokinetics-pharmacodynamics of pyrazinamide in a novel in vitro model of tuberculosis for sterilizing effect: a paradigm for faster assessment of new antituberculosis drugs
Abstract
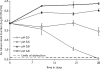
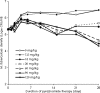
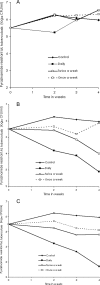
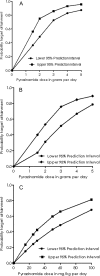
There are currently renewed efforts to develop drugs that could shorten the duration of antituberculosis therapy. This is best achieved by optimizing the sterilizing effect. However, the current pathway for the development of new molecules with the potential to have a sterilizing effect is inefficient. We designed an in vitro pharmacokinetic-pharmacodynamic model in which Mycobacterium tuberculosis replicating slowly at pH 5.8 was exposed to pyrazinamide by use of the concentration-time profiles encountered in patients. The sterilizing effect rates and the time to the emergence of drug resistance were examined. Daily pyrazinamide dosing for 28 days accurately achieved (i) the pyrazinamide pharmacokinetic parameters, (ii) the lack of early bactericidal activity, (iii) a sterilizing effect rate of 0.10 log(10) CFU/ml per day starting on day 6 of therapy, and (iv) a time to the emergence of resistance of the from 2 to 3 weeks of monotherapy encountered in patients with tuberculosis. Next, dose-scheduling studies were performed. The sterilizing effect was linked to the pyrazinamide ratio of the area under the concentration-time curve from 0 to 24 h (AUC(0-24)) to the MIC (r(2) = 0.80 to 0.90), with 90% of the maximal effect being achieved by an AUC(0-24)/MIC of 209.08. Resistance suppression was associated with the percentage of time that the concentration persisted above the MIC (r(2) = 0.73 to 0.91). Monte Carlo simulations of 10,000 patients demonstrated that the currently recommended pyrazinamide doses (15 to 30 mg/kg of body weight/day) achieved the AUC(0-24)/MIC of 209.08 in the epithelial lining fluid of only 15.1 to 53.3% of patients. Doses of >60 mg/kg per day performed better. Our vitro model for the sterilizing effect, together with Monte Carlo simulations, can be used for the faster identification of the clinical doses that are needed to achieve a sterilizing effect and that can then be studied in clinical trials.
Figures
References
-
- Aly, S., K. Wagner, C. Keller, S. Malm, A. Malzan, S. Brandau, F. C. Bange, and S. Ehlers. 2006. Oxygen status of lung granulomas in Mycobacterium tuberculosis-infected mice. J. Pathol. 210:298-305. - PubMed
-
- Anonymous. 1981. Controlled trial of four thrice-weekly regimens and a daily regimen all given for 6 months for pulmonary tuberculosis. Lancet i:171-174. - PubMed
-
- Blaser, J., B. B. Stone, and S. H. Zinner. 1985. Two compartment kinetic model with multiple artificial capillary units. J. Antimicrob. Chemother. 15(Suppl. A):131-137. - PubMed
-
- Blumberg, H. M., W. J. Burman, R. E. Chaisson, C. L. Daley, S. C. Etkind, L. N. Friedman, P. Fujiwara, M. Grzemska, P. C. Hopewell, M. D. Iseman, R. M. Jasmer, V. Koppaka, R. I. Menzies, R. J. O'Brien, R. R. Reves, L. B. Reichman, P. M. Simone, J. R. Starke, and A. A. Vernon. 2003. American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America: treatment of tuberculosis. Am. J. Respir. Crit. Care Med. 167:603-662. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

