The molecular effects of skeletal muscle myosin regulatory light chain phosphorylation
- PMID: 19458282
- PMCID: PMC2724231
- DOI: 10.1152/ajpregu.00171.2009
The molecular effects of skeletal muscle myosin regulatory light chain phosphorylation
Abstract
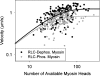
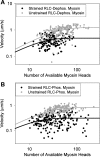
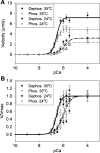
Phosphorylation of the myosin regulatory light chain (RLC) in skeletal muscle has been proposed to act as a molecular memory of recent activation by increasing the rate of force development, ATPase activity, and isometric force at submaximal activation in fibers. It has been proposed that these effects stem from phosphorylation-induced movement of myosin heads away from the thick filament backbone. In this study, we examined the molecular effects of skeletal muscle myosin RLC phosphorylation using in vitro motility assays. We showed that, independently of the thick filament backbone, the velocity of skeletal muscle myosin is decreased upon phosphorylation due to an increase in the myosin duty cycle. Furthermore, we did not observe a phosphorylation-dependent shift in calcium sensitivity in the absence of the myosin thick filament. These data suggest that phosphorylation-induced movement of myosin heads away from the thick filament backbone explains only part of the observed phosphorylation-induced changes in myosin mechanics. Last, we showed that the duty cycle of skeletal muscle myosin is strain dependent, consistent with the notion that strain slows the rate of ADP release in striated muscle.
Figures

Similar articles
-
Myosin regulatory light chain phosphorylation enhances cardiac β-myosin in vitro motility under load.Arch Biochem Biophys. 2015 Aug 15;580:14-21. doi: 10.1016/j.abb.2015.06.014. Epub 2015 Jun 25. Arch Biochem Biophys. 2015. PMID: 26116789 Free PMC article.
-
Phosphorylation of the regulatory light chains of myosin affects Ca2+ sensitivity of skeletal muscle contraction.J Appl Physiol (1985). 2002 Apr;92(4):1661-70. doi: 10.1152/japplphysiol.00858.2001. J Appl Physiol (1985). 2002. PMID: 11896035
-
Comparison of orientation and rotational motion of skeletal muscle cross-bridges containing phosphorylated and dephosphorylated myosin regulatory light chain.J Biol Chem. 2013 Mar 8;288(10):7012-23. doi: 10.1074/jbc.M112.434209. Epub 2013 Jan 14. J Biol Chem. 2013. PMID: 23319584 Free PMC article.
-
Myosin light chain kinase and the role of myosin light chain phosphorylation in skeletal muscle.Arch Biochem Biophys. 2011 Jun 15;510(2):120-8. doi: 10.1016/j.abb.2011.01.017. Epub 2011 Feb 1. Arch Biochem Biophys. 2011. PMID: 21284933 Free PMC article. Review.
-
Signaling to myosin regulatory light chain in sarcomeres.J Biol Chem. 2011 Mar 25;286(12):9941-7. doi: 10.1074/jbc.R110.198697. Epub 2011 Jan 21. J Biol Chem. 2011. PMID: 21257758 Free PMC article. Review.
Cited by
-
Regulatory light chain phosphorylation augments length-dependent contraction in PTU-treated rats.J Gen Physiol. 2019 Jan 7;151(1):66-76. doi: 10.1085/jgp.201812158. Epub 2018 Dec 6. J Gen Physiol. 2019. PMID: 30523115 Free PMC article.
-
Blebbistatin Effects Expose Hidden Secrets in the Force-Generating Cycle of Actin and Myosin.Biophys J. 2018 Jul 17;115(2):386-397. doi: 10.1016/j.bpj.2018.05.037. Biophys J. 2018. PMID: 30021113 Free PMC article.
-
Pseudophosphorylation of cardiac myosin regulatory light chain: a promising new tool for treatment of cardiomyopathy.Biophys Rev. 2017 Feb;9(1):57-64. doi: 10.1007/s12551-017-0248-8. Epub 2017 Jan 25. Biophys Rev. 2017. PMID: 28510043 Free PMC article. Review.
-
Mechanical properties of respiratory muscles.Compr Physiol. 2013 Oct;3(4):1553-67. doi: 10.1002/cphy.c130003. Compr Physiol. 2013. PMID: 24265238 Free PMC article. Review.
-
The direct molecular effects of fatigue and myosin regulatory light chain phosphorylation on the actomyosin contractile apparatus.Am J Physiol Regul Integr Comp Physiol. 2010 Apr;298(4):R989-96. doi: 10.1152/ajpregu.00566.2009. Epub 2010 Jan 20. Am J Physiol Regul Integr Comp Physiol. 2010. PMID: 20089714 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

