Multipotent adult progenitor cells can suppress graft-versus-host disease via prostaglandin E2 synthesis and only if localized to sites of allopriming
- PMID: 19458354
- PMCID: PMC2713464
- DOI: 10.1182/blood-2009-03-213850
Multipotent adult progenitor cells can suppress graft-versus-host disease via prostaglandin E2 synthesis and only if localized to sites of allopriming
Abstract
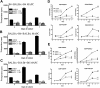
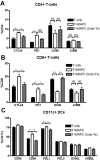
Multipotent adult progenitor cells (MAPCs) are nonhematopoietic stem cells capable of giving rise to a broad range of tissue cells. As such, MAPCs hold promise for tissue injury repair after transplant. In vitro, MAPCs potently suppressed allogeneic T-cell activation and proliferation in a dose-dependent, cell contact-independent, and T-regulatory cell-independent manner. Suppression occurred primarily through prostaglandin E(2) synthesis in MAPCs, which resulted in decreased proinflammatory cytokine production. When given systemically, MAPCs did not home to sites of allopriming and did not suppress graft-versus-host disease (GVHD). To ensure that MAPCs would colocalize with donor T cells, MAPCs were injected directly into the spleen at bone marrow transplantation. MAPCs limited donor T-cell proliferation and GVHD-induced injury via prostaglandin E(2) synthesis in vivo. Moreover, MAPCs altered the balance away from positive and toward inhibitory costimulatory pathway expression in splenic T cells and antigen-presenting cells. These findings are the first to describe the immunosuppressive capacity and mechanism of MAPC-induced suppression of T-cell alloresponses and illustrate the requirement for MAPC colocalization to sites of initial donor T-cell activation for GVHD inhibition. Such data have implications for the use of allogeneic MAPCs and possibly other immunomodulatory nonhematopoietic stem cells for preventing GVHD in the clinic.
Figures

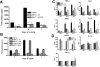



Similar articles
-
Human multipotent adult progenitor cells effectively reduce graft-vs-host disease while preserving graft-vs-leukemia activity.Stem Cells. 2021 Nov;39(11):1506-1519. doi: 10.1002/stem.3434. Epub 2021 Jul 28. Stem Cells. 2021. PMID: 34255899 Free PMC article.
-
Clinical-Grade Human Multipotent Adult Progenitor Cells Block CD8+ Cytotoxic T Lymphocytes.Stem Cells Transl Med. 2016 Dec;5(12):1607-1619. doi: 10.5966/sctm.2016-0030. Epub 2016 Jul 27. Stem Cells Transl Med. 2016. PMID: 27465071 Free PMC article.
-
Multipotent Adult Progenitor Cells Suppress T Cell Activation in In Vivo Models of Homeostatic Proliferation in a Prostaglandin E2-Dependent Manner.Front Immunol. 2018 Apr 23;9:645. doi: 10.3389/fimmu.2018.00645. eCollection 2018. Front Immunol. 2018. PMID: 29740426 Free PMC article.
-
Multipotent adult progenitor cells: an update.Novartis Found Symp. 2005;265:55-61; discussion 61-5, 92-7. Novartis Found Symp. 2005. PMID: 16050250 Review.
-
Alloreactivity as therapeutic principle in the treatment of hematologic malignancies. Studies of clinical and immunologic aspects of allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning.Dan Med Bull. 2007 May;54(2):112-39. Dan Med Bull. 2007. PMID: 17521527 Review.
Cited by
-
Concise review: hitting the right spot with mesenchymal stromal cells.Stem Cells. 2010 Aug;28(8):1446-55. doi: 10.1002/stem.459. Stem Cells. 2010. PMID: 20597105 Free PMC article. Review.
-
Suppression of IL-7-dependent Effector T-cell Expansion by Multipotent Adult Progenitor Cells and PGE2.Mol Ther. 2015 Nov;23(11):1783-1793. doi: 10.1038/mt.2015.131. Epub 2015 Jul 28. Mol Ther. 2015. PMID: 26216515 Free PMC article.
-
A Preliminary Investigation towards the Risk Stratification of Allogeneic Stem Cell Recipients with Respect to the Potential for Development of GVHD via Their Pre-Transplant Plasma Lipid and Metabolic Signature.Cancers (Basel). 2019 Jul 25;11(8):1051. doi: 10.3390/cancers11081051. Cancers (Basel). 2019. PMID: 31349646 Free PMC article.
-
Neuroinflammatory signals enhance the immunomodulatory and neuroprotective properties of multipotent adult progenitor cells.Stem Cell Res Ther. 2015 Sep 16;6(1):176. doi: 10.1186/s13287-015-0169-z. Stem Cell Res Ther. 2015. PMID: 26377390 Free PMC article.
-
Multipotent adult progenitor cells induce regulatory T cells and promote their suppressive phenotype via TGFβ and monocyte-dependent mechanisms.Sci Rep. 2021 Jun 30;11(1):13549. doi: 10.1038/s41598-021-93025-x. Sci Rep. 2021. PMID: 34193955 Free PMC article.
References
-
- Fibbe WE, Nauta AJ, Roelofs H. Modulation of immune responses by mesenchymal stem cells. Ann N Y Acad Sci. 2007;1106:272–278. - PubMed
-
- Uccelli A, Moretta L, Pistoia V. Immunoregulatory function of mesenchymal stem cells. Eur J Immunol. 2006;36:2566–2573. - PubMed
-
- Rasmusson I, Ringden O, Sundberg B, Le Blanc K. Mesenchymal stem cells inhibit lymphocyte proliferation by mitogens and alloantigens by different mechanisms. Exp Cell Res. 2005;305:33–41. - PubMed
-
- Di Nicola M, Carlo-Stella C, Magni M, et al. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood. 2002;99:3838–3843. - PubMed
-
- Meisel R, Zibert A, Laryea M, Gobel U, Daubener W, Dilloo D. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase–mediated tryptophan degradation. Blood. 2004;103:4619–4621. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

